Riesenkauz (Strix sp.)
Diese rätselhafte Form ist nur anhand eines einzigen Fossils (USNM 214769) bekannt, einer Unterkiefersymphyse (Symphysis mandibulae) also quasi einem Unterschnabel, der sich einer Eulenart zuordnen lässt.
Das Fossil ähnelt am ehesten dem entsprechenden Knochen in Eulen der Gattung Strix, stammt aber von einer sehr viel größeren Art, viel größer als selbst die größte Art dieser Gattung, dem Bartkauz (Strix nebulosa Forster) und auch größer als jede andere lebende Eulenart; der Vogel mag zu Lebzeiten eine Gesamtlänge von etwas über 1 m erreicht haben! [1]
***
Die ersten Menschen, die im Oberen Pleistozän nach Nordamerika einwanderten, sind sehr wahrscheinlich Augenzeugen dieser riesigen Eulenart geworden. Und tatsächlich existieren Überlieferungen diverser Gruppen nordamerikanischen Unreinwohner, die von riesigen Eulen berichten, die sehr gefährlich gewesen sein- und sogar Jagd auf Kinder gemacht haben sollen.
*********************
Quelle:
[1] Storrs L. Olson: A very large enigmatic owl (Aves: Strigidae) from the Late Pleistocene at Ladds, Georgia. H. H. Genoways & M. R. Dawson: Contributions in Quaternary vertebrate paleontology: A volume in memorial to John E. Guilday. Carnegie Museum of Natural History Special Publication 8: 44-46. 1984
*********************
bearbeitet: 23.04.2025
Author Archives: Alexander Lang
Grünracke
Grünracke (kein wissenschaftlicher Name)
Diese Form, die von irgendwo in Indien stammen soll, ist nur anhand der Beschreibung und der dazugehörigen Darstellung von François Le Vaillant aus dem Jahr 1806 bekannt; die Beschreibung lässt einige Fragen offen und erscheint nicht hundertprozentig mit der Abbildung übereinzustimmen.
“Voici encore une espèce nouvelle, qui paroît se rapprocher beaucoup du rollier vulgaire, mais dont il diffère cependant assez pour former une espèce distincte et séparée. Nous ne connoissons pas le pays natal de cet oiseau , du moins le canton de l’Inde d’où il a été importé en Europe par M. Poivre, ….
Ce rollier, que nous appelons verd, parceque le verd est en effet la couleur dominante de son plumage, a tous les caractères et toutes les formes du rollier vulgaire, dont nous donnons la description dans l’article suivant; mais il est un peu moins fort de taille que lui, comme on le verra en comparant les figures de grandeur naturelle que nous donnons de l’un et de l’autre: il a les plumes du front jusqu’aux yeux, ainsi que celles qui avoisinent la base du bec et la gorge, d’un blanc roussâtre: la tête, le cou, le haut du dos, toutes les plumes scapulaircs, les plumes alaires les plus proches du corps, et généralement toutes leurs couvertures supérieures sont d’un verd aiguë-marine: toutes les plumes du dessous du corps, depuis le blanc roussâtre de la gorge jusqu’au basventre, sont aussi verd aigue-marine, mais d’un ton plus clair, et qui, prenant une nuance blanche vers le bas-ventre , devient enfin d’un blanc légèrement teint du même verd sur les couvertures du dessous de la queue: les six premières grandes pennes alaires sont d’un beau bleu violâtre; les suivantes sont de plus légèrement bordées de verd à leurs pointes: les plumes du croupion et les couvertures du dessus de la queue sont d’un verd bleuâtre, ainsi que les deux plumes intermédiaires de celle-ci, bleues par-tout ailleurs tant en-dessus qu’en-dessous, où ce bleu est cependant un peu plus clair: le bec est noir, et les pieds sont roux.” [1]
Übersetzung:
“Hier ist eine weitere neue Art, die der Blauracke sehr nahe zu sein scheint, von der sie sich aber dennoch so weit unterscheidet, dass sie eine eigene und eigenständige Art bildet. Wir kennen das Heimatland dieses Vogels nicht, zumindest nicht den Kanton in Indien, von wo aus er von Herrn Poivre, … importiert wurde.
Diese Racke, die wir Grün nennen, weil Grün tatsächlich die vorherrschende Farbe ihres Gefieders ist, hat alle Charaktere und alle Formen der gewöhnlichen Racke [Blauracke (Coracias garrulus L.)], die wir im folgenden Artikel beschreiben; aber sie ist etwas kleiner als sie, wie wir sehen werden, wenn wir die natürlichen Größenangaben vergleichen, die wir von beiden geben: die Federn von der Stirn bis zu den Augen sowie die, die die Basis des Schnabels umgeben, sind rötlich weiß: der Kopf, der Hals, der obere Rücken, alle Schulterfedern, die dem Körper am nächsten liegenden Flügelfedern und im Allgemeinen alle ihre oberen Bedeckungen sind von kräftigem Aquamaringrün: alle Federn auf der Unterseite des Körpers, vom rötlichen Weiß der Kehle bis zum Unterbauch, sind ebenfalls aquamaringrün, jedoch von einem helleren Ton, und nehmen zum Unterbauch hin einen weißen Farbton an und werden schließlich weiß, leicht mit demselben Grün gefärbt auf den Decken der Schwanzunterseite: die ersten sechs großen Flügelfedern sind von einem wunderschönen Purpurblau; die folgenden sind an ihren Spitzen heller mit Grün umrandet: die Federn des Bürzels und die Federn an der Oberseite des Schwanzes sind von bläulichem Grün, die beiden Mittelfedern desselben sind überall blau, sonst beide oben als unten, wo dieses Blau allerdings etwas heller ist: Der Schnabel ist schwarz und die Füße sind rot. “

(not in copyright)
Es handelt sich hierbei ziemlich wahrscheinlich um eine der vielen künstlichen Chimären, aus den Einzelteilen verschiedener Vogelarten zusammengebauten Stopfpräparaten, wie sie im frühen 19.Jahrhundert in vielen Sammlungen zu finden waren – je absonderlicher diese ausfielen umso höher waren die Preise zu denen geneigte Sammler sie zu kaufen tatsächlich bereit waren.
*********************
Quelle:
[1] François Le Vaillant: Histoire naturelle des oiseaux de paradis et des rolliers: suivie de celle des toucans et des barbus. Vol. 1. Paris, chez Denné le jeune, Libraire, rue Vivienne, N°, 10. & chez Perlet, Libraire, rue de Tournon 1806
*********************
bearbeitet: 23.04.2024
Dombeys Motmot
Dombeys Motmot (Baryphthengus dombeyi (Lesson))
Diese kaum bekannte Form ist nur anhand der Beschreibung (und Darstellung) von François Le Vaillant aus dem Jahr 1806 bekannt, die offenbar auf zwei Exemplaren beruht, die nicht mehr existieren; diese Beschreibung ist äußerst langatmig, daher werde ich sie hier nicht vollständig wiedergeben.
Der Vogel soll aus Peru stammen und dort in der Umgebung der heutigen Hauptstadt Lima vorgekommen sein.
“Nous n’étendrons pas davantage la description du momot dombé, parceque la figure exacte que nous en avons donnée mettra le lecteur qui le comparera à celui de l’espèce précédente, à même de saisir les rapports et les différences qui existent entre eux. Le houtou se trouve à la Guyane, et le momot dombé habite les forêts des environs de Lima, où le voyageur que j’ai nommé m’a assuré qu’il étoit très commun, et qu’il n’avoit remarqué aucune différence entre beaucoup d’individus qu’il avoit vus de l’espèce et les deux qu’il avoit rapportés, dont l’un fut déposé avec plusieurs autres beaux oiseaux du Pérou au cabinet du roi. Il est fâcheux que cet individu ait été entièrement détruit par les fumigations de soufre et les insectes: quant à l’autre nous ne savons ce qu’il est devenu.” [1]
Übersetzung:
“Wir werden die Beschreibung des Dombeys Motmots nicht weiter ausdehnen, da die genaue Abbildung, die wir wiedergegeben haben, den Leser, der sie mit denen der vorhergehenden Arten vergleicht, in die Lage versetzen wird, die Beziehungen und Unterschiede zwischen ihnen zu verstehen. Der Houtou [Braunscheitelmotmot (Momotus mexicanus Swainson)] kommt in Guyana vor, und Dombeys Motmot bewohnt die Wälder der Umgebung von Lima, wo der Reisende, den ich nannte, mir versicherte, dass er sehr häufig sei und dass er zwischen vielen Individuen, die er gesehen hatte, keinen Unterschied bemerkt habe und von den beiden, die er zurückgebracht hatte, einer zusammen mit mehreren anderen schönen Vögeln Perus im Kabinett des Königs deponiert wurde. Es ist bedauerlich, dass dieses Individuum durch die Begasung mit Schwefel und durch Insekten vollständig zerstört wurde; was den anderen betrifft, wissen wir nicht, was daraus geworden ist.“

(not in copyright)
*********************
Quellen:
[1] François Le Vaillant: Histoire naturelle des oiseaux de paradis et des rolliers: suivie de celle des toucans et des barbus. Vol. 1. Paris, chez Denné le jeune, Libraire, rue Vivienne, N°, 10. & chez Perlet, Libraire, rue de Tournon 1806
[2] Julian P. Hume: Extinct Birds. Bloomsbury Natural History; 2nd edition 2017
*********************
bearbeitet: 23.04.2024
Bensbachs Paradiesvogel
Bensbachs Paradiesvogel (Janthothorax bensbachi Büttikofer)
Diese Form wurde 1895 als neue Art beschrieben und ist bis heute nur anhand eines einzigen Exemplars bekannt, das in den Arfak-Bergen im Nordosten der Vogelkop-Halbinsel im Westen Neuguineas gefunden wurde.
Tatsächlich handelt es sich hierbei um einen Hybrid aus zwei sehr gut bekannten Paradiesvogelarten, nämlich dem Kleinen Paradiesvogel (Paradisaea minor Shaw) und dem Prachtparadiesvogel (Ptiloris magnificus(Viellot)).

(public domain)
*********************
Quellen:
[1] Errol Fuller: The Lost Birds of Paradise. Airlife 1996
[2] Clifford B. Frith; Bruce M. Beehler: The Birds of Paradise: Paradisaeidae. Oxford University Press 1998
*********************
bearbeitet: 23.04.2024
Drüsentaube
Drüsentaube (Columba carunculata Temminck)
Dies ist eine weitere von zahlreichen mysteriösen Vogelarten, die nur anhand von zeitgenössischen Beschreibungen und den dazugehörigen Darstellungen bekannt sind.

(public domain)
Die Originalbeschreibung stammt aus dem Jahr 1809 und ist mir nicht zugänglich, daher greife ich auf eine Beschreibung aus dem Jahr 1839 zurück, die sogar in deutscher Sprache verfasst wurde.:
“Von allen bis jetzt bekannten Tauben verbindet sich nach Temminck keine so enge wie diese mit den eigentlichen Hühnern, sowohl in der äußeren Erscheinung als in Gewohnheiten und Lebensweise, und auffallend dahin deutend ist gleich beim ersten Anblick das fleischige, drüsenartige Gehänge, wie wir solches and unserem Haushahn sehen. Die Drüsentaube ist wieder in Südafrika heimisch; sie wurde von Le Vaillant zuerst in der Gegend von Numaqua [Namibia] entdeckt, und folgenden näheren Bericht hat dieser thätige, gelehrte Naturforscher darüber in seinem schönen Werke: “Ueber die afrikanischen Vögel” ertheilt.
“Ihr Zusammengehören mit den Tauben … erkennt man zuerst an der Form des Schnabels, der genau mit diesen übereinstimmt, dann in den Eigenschaften und der Lage des Gefieders; sehr verschieden ist sie aber durch den rothen, nackten, fleischigen Auswuchs am Halse, dann durch die längeren Füße, einen runden plumpen Leib, durch das abhängige Tragen des Schwanzes, ungefähr wie beim Repphuhne, und endlich durch ihre mehr gerundeten Flügel, alles sonstige Kennzeichen der Hühnerarten, so daß sie am besten zwischen diesen und den Tauben zur Verbindung Beider steht. Sie baut ihr Nest in leichte Gruben auf der Erde aus dünnen Zweigen und dürren Halmen; das Weibchen legt sechs bis acht röthlich weiße Eier, die wechselnd von ihm und dem Männchen gebrütet werden. Die Jungen haben nach dem Auskriechen eine Bekleidung von röthlich grauem Flaume, laufen sogleich den Eltern nach, die sie fortdauernd durch einen sonderbaren Ruf zusammenhalten und mit ausgebreiteten Flügeln vor der kalten Nachtluft schützen. Ihre erste Nahrung besteht aus Ameiseneiern, kleinen Insecten und Würmern, welche von den Eltern ausgesucht und ihnen überlassen werden. Bei zunehmender Größe müssen sie selbst für sich sorgen, dann sind ihnen Getreide, Samenkörner, Beeren und Insecten willkommen. Sie bleiben, wie die Repphühner, in Flügen beisammen und trennen sich nur paarweise um die Brütezeit.”
In der Größe gleicht die Drüsentaube unserer gewöhnlichen Turteltaube, nur ist der Rumpf dicker und runder. Die Basis des Schnabels und der Vorderkopf werden von einer nackten, lebhaft rothen Haut überzogen, und am Halse hängt eine gleichfarbige Substanz, die sich zu beiden Seiten gegen die Ohren zieht. Kopf, Hals und Brust sind purpurgrau, Mantel, Achseln und Flügeldecken bleichgrau die Federn weiß gesäumt. Der Bauch, der hintere und Ober- und Untertheil der Schwanzfedern, so wie die Seiten unter den Flügeln, sind rein weiß. Der Schwanz kurz, abgerundet, die Federn tief rothbraun, mit Ausnahme der ersten Feder auf jeder Seite, deren äußere Fahne weiß ist. Der Schnabel zeigt sich an der Wurzel röthlich, gegen die Spitze schwarz. Die Füße sind purpurroth, mit sechseckigen Schuppen. Die Iris hat einen doppelten, inwendig gelben, auswendig rothen Kreis. Das Weibchen gleicht dem Männchen in der Lage des Gefieders, aber die Färbung desselben ist trüber und der Hals nicht, wie dort, behängt.” [1]
Interessant sind die Hinweise auf die Küken der ‘Art’, die hier als Nestflüchter beschrieben werden, welche gleich nach dem Schlüpfen herumlaufen und von den Erwachsenen mit Insektennahrung versorgt werden – tatsächlich sind sämtliche Taubenarten Nesthocker und werden obendrein vom Weibchen mit so genannter Kropfmilch gefüttert ….

(public domain)
Die Drüsentaube gilt heute als eine der vielen frei erfundenen Vogelarten, die vor allem im frühen- bis mittleren 19. Jahrhundert in der Literatur sowie in Sammlungen seltener (aus Einzelteilen diverser Vögel zusammengebastelter) Vogelpräparate auftauchen.:
“Columba Carunculata Temminck, 1809: 19, pl. 11 is not applicable, artefact.
The name was based on an artefact. Temminck (1809) referred to a single specimen in the collection of Levaillant.” [2]
Übersetzung:
“Columba Carunculata Temminck, 1809: 19, Taf. 11 ist nicht zuordenbar, Artefakt.
Der Name basierte auf einem Artefakt. Temminck (1809) verwies auf ein einziges Exemplar in der Sammlung von Levaillant.“
*********************
Quellen:
[1] Friedrich Treitschke: Naturgeschichte der Tauben, nach Prideaux Selby. Naturgeschichtliches Cabinet des Thierreiches VII. Pesth 1839
[2] Steven D. van der Mije; Pepijn Kamminga; René W. R. J. Dekker: Type specimens of non-passerines in Naturalis Biodiversity Center (Animalia, Aves). Zookeys 1155: 1-311. 2023
*********************
edited: 19.04.2024
Neue Art – El Dorado Ameisenpitta (Grallaria sp. ‘El Dorado’)
Diese vermutlich neue Vogelart wurde bereits 2015 entdeckt (zu dieser Zeit aber nicht als neu erkannt), so dass die offizielle Entdeckung als neue Art tatsächlich erst im Jahr 2024 stattfand. [1]
Der El Dorado Ameisenpitta lebt im El Dorado Nature Reserve in der Sierra Nevada de Santa Marta im Norden Kolumbiens, eine Region in der sehr viele endemische Vogelformen beheimatet sind.
Ursprünglich wurden die Vögel als Schuppenbauch-Ameisenpittas (Grallaria squamigera Prévost & Des Murs) identifiziert; neuerdings geht man jedoch davon aus, dass sie eine eigenständige Art darstellen könnten, vor allem aufgrund ihrer Lautäußerungen, die offenbar deutlich anders klingen als die des Schuppenbauch-Ameisenpittas.
Noch ist nicht restlos geklärt, ob es sich tatsächlich um eine neue Art oder Unterart handelt, und endgültige Klarheit wird sicher erst durch DNA-Untersuchungen zu schaffen sein, es ist jedoch bereits klar, dass die Familie der Ameisenpittas tatsächlich weitaus artenreicher ist als zuvor gedacht. [2]

Photo: Fundacion ProAves
https://www.inaturalist.org/people/fundacion_proaves
http://creativecommons.org/licenses/by-nc/4.0/

Photo: Norma Malinowski
https://www.inaturalist.org/people/bogwalker
https://creativecommons.org/licenses/by/4.0/
*********************
Quellen:
[1] Sophie A. H. Osborn; Chad V. Olson: New record and known-range extension of Undulated Antpitta (Grallaria squamigera) in the Sierra Nevada de Santa Marta of Colombia. Boletín SAO 24(1&2): 9-12. 2015
[2] Morton L. Isler; R. Terry Chesser; Mark B. Robbins; Andrés M. Cuervo; Carlos Daniel Cadena; Peter A. Hosner: Taxonomic evaluation of the Grallaria rufula (Rufous Antpitta) somplex (Aves: Passeriformes: Grallariidae) distinguishes sixteen species. Zootaxa 4817(1): 1-74. 2020
*********************
bearbeitet: 18.04.2024
Türkentaube

*********************
bearbeitet: 09.04.2024
Fossile Anseriformes
Familie incertae sedis
Conflicto antarcticus Tambussi et al.
Eoneornis australis Ameghino
Eutelornis patagonicus Ameghino
Kookne yeutensis Novas et. al. (?)
Naranbulagornis khun Zelenkov
„Oxyura“ doksana Mlíkovský
Paakniwatavis grandei Musser & Clarke
Palaeopapia eous Harrison & Walker
Palaeopapia hamstaeadiensis Harrison & Walker
Paracygnopterus scotti Harrison & Walker
Paranyroca magna Miller & Compton
Peioa australis Agnolín
Petropluvialis simplex Harrison & Walker
Wuketru howardae (Cracraft)
Anachronornithidae
Anachronornis anhimops Houde et al.
Danielsavis nazensis Houde et al.
Anatidae
Afrocygnus chauvireae Louchart, Vignaud, Likius, Mackaye & Brunet
Aix praeclara Zelenkoy & Kurochkin
Aldabranas cabri Harrison & Walker
Allgoviachen tortonica Mayr, Lechner & Böhme [7]
Aminornis excavatus Ameghino
Anabernicula gracilenta Ross
Anabernicula oregonensis Howard
Anas amotape Campbell
Anas apscheronica Burchak-Abramovich
Anas basaltica Bayer
Anas crassa Milne-Edwards
Anas isarensis Lambrecht
Anas itchtucknee McCoy
Anas kurochkini Zelenkov & Panteleyev
Anas luederitzensis Lambrecht
Anas macroptera Milne-Edwards
Anas meyeri Milne-Edwards
Anas pachyscelus Wetmore
Anas risgoviensis von Ammon
Anas robusta Milne-Edwards
Anas sanctaehelenae Campbell
Anas schneideri Emslie
Anas skalicensis Bayer
Anas soporata Kurochkin
Anas talarae Campbell
Ankonetta larriestrai Cenizo & Agnolín
Annakacygna hajimei Matsuoka & Hasegawa [5]
Annakacygna yoshiiensis Matsuoka & Hasegawa [5]
Anser arenosus Bickart
Anser arizonae Bickart
Anser atavus Fraas
Anser azerbaidzhanicus Serebrovskii
Anser cygniformis Fraas
Anser devjatkini Kuročkin
Anser djuktaiensis Zelenkov & Kurochkin [4]
Anser eldaricus Burchak-Abramovich & Gadzyev
Anser oeningensis (Meyer)
Anser pratensis (Short)
Anser pressus (Brodkorb)
Anser tchikoicus Kuročkin
Anser thompsoni Martin & Mengel
Anser thraceiensis Burchak-Abramovich & Nikolov
Anserobranta robusta (Milne-Edwards)
Anserobranta tarabukinii (Kurochkin & Ganea)
Archaeocycnus lacustris De Vis
Australotadorna alecwilsoni Worthy
Aythya denesi (Kessler)
Branta dickeyi Miller
Branta esmeralda (Burt)
Branta howardae Miller
Branta hypsibata (Cope)
Branta propinqua Shufeldt
Branta thessaliensis Boev & Koufos
Branta woolfendeni Bickart
Caerulonettion natator (Milne-Edwards)
Cayaoa bruneti Tonni
Chendytes milleri Howard
Chenoanas asiatica Zelenkov et al.
Chenoanas deserta Zelenkov
Chenoanas sansaniensis (Milne-Edwards)
Chenopis nanus De Vis
Cousteauvia kustovia Zelenkov [1]
Cygnavus formosus Kurochkin
Cygnavus senckenbergi Lambrecht
Cygnopterus affinis (Van Beneden)
Cygnus atavus (Fraas)
Cygnus csakvarensis Lambrecht
Cygnus equitum Bate
Cygnus falconeri Parker
Cygnus hibbardi Brodkorb
Cygnus lacustris (De Vis)
Cygnus liskunae (Kuročkin)
Cygnus mariae Bickart
Cygnus paloregonus Cope
Cygnus pristinus Kurochkin
Cygnus sp. ‘Dursunlu, Turkey’
Cygnus verae Boev
Dendrochen robusta Miller
Dunstanetta johnstoneorum Worthy, Tennyson, Jones, McNamara & Douglas
Eonessa anaticula Wetmore
Eremochen russelli Brodkorb
Garganornis ballmanni H. J. M. Meyer
Guguschia nailiae Aslanova & Burczak-Abramovicz
Kaikenia mourerchauvirea Agnolín
Lavanttalornis hassleri Bocheński et al.
Loxornis clivus Ameghino
Manuherikia douglasi Worthy, Tennyson, Hand & Scofield
Manuherikia lacustrina Worthy, Tennyson, Jones, McNamara & Douglas
Manuherikia minuta Worthy, Tennyson, Jones, McNamara & Douglas
Manuherikia primadividua Worthy et al. [2]
Matanas enrighti Worthy, Tennyson, Jones, McNamara & Douglas
Megalodytes morejohni Howard
Mergellus mochanovi Zelenkov & Kurochkin
Mionetta blanchardi (Milne-Edwards)
Mionetta consobrina (Milne-Edwards)
Mionetta defossa Selenkow
Mionetta eversa (Wetmore)
Mioquerquedula palaeotagaica Zelenkov
Miotadorna sanctibathansi Worthy, Tennyson, Jones, McNamara & Douglas
Nettapus anatoides Depéret
Nogusunna conflictoides Zelenkov
Notochen bannockburnensis Worthy et al. [6]
Oxyura bessomi Howard
Oxyura hulberti Emslie
Oxyura zapatanima Alvarez
Pinpanetta fromensis Worthy
Pinpanetta tedfordi Worthy
Pinpanetta vickersrichae Worthy
Protomelanitta bakeri Stidham & Zelenkov
Protomelanitta gracilis Zelenkov
Romainvillia kazakhstanensis Zelenkov
Romainvillia stehlini Ledebinsky
Saintandrea chenoides Mayr & De Pietri
Selenonetta lacustrina Zelenkov
Sharganetta mongolica Zelenkov
Sibirionetta formozovi Zelenkov
Sinanas diatomas Yeh
Spatula praeclypeata Zelenkov
Tadorna petrina Zelenkov
Tagayanetta palaeobaikalensis Zelenkov
Tamtamia yzurietai Agnolín
Telornis impressus Ameghino
Tirarinetta kanuka Worthy
Anhimidae
Chainkanas koshon Agnolín
Chaunoides antiquus Alvarenga
Anseranatidae
Anatalavix oxfordi Olson
Anatalavis rex (Shufeldt)
Anserpica kiliani Mourer-Chauviré
Eoanseranas handae Worthy & Scanlon
Brontornithidae
Brontornis burmeisteri Moreno & Mercerat
Dromornithidae
Bullockornis planei Rich
Dromornis australis Owen
Dromornis murrayi Worthy, Handley, Archer & Hand
Dromornis stirtoni Rich
Genyornis newtoni Stirling & Zietz
Ilbandornis lawsoni Rich
Ilbandornis woodburnei Rich
Presbyornithidae
Bumbalavis anatoides Zelenkov [3]
Haedonornis hantoniensis Harrison & Walker
Murgonornis archeri Worthy et al.
Presbyornis isoni Olson
Presbyornis mongoliensis Kurochkin & Dyke
Presbyornis pervetus Wetmore
Presbyornis recurvirostrus Hardy
Telmabates antiquus Howard
Teviornis gobiensis Kurochkin et al.
Wilaru prideauxi De Pietri et al.
Wilaru tedfordi Boles et al.
Zhylgaia aestiflua Nessov
*********************
Quellen:
[1] Nikita Zelenkov: The oldest diving anseriform bird from the late Eocene of Kazakhstan and the evolution of aquatic adaptations in the intertarsal joint of waterfowl. Acta Palaeontologica Polonica 65 (4): 733-742. 2020
[2] Trevor H. Worthy; R. Paul Scofield; Steven W. Salisbury; Suzanne J. Hand; Vanesa L. De Pietri; Jacob C. Blokland; Michael Archer: A new species of Manuherikia (Aves: Anatidae) provides evidence of faunal turnover in the St Bathans fauna, New Zealand. Geobios. https://doi.org/10.1016/j.geobios.2021.08.002. 2021
[3] Nikita V. Zelenkov: A revision of the Paleocene-Eocene Mongolian Presbyornithidae (Aves: Anseriformes). Paleontological Journal 55(3): 83-91. 2021
[4] N. V. Zelenkov; G. G. Boeskorov: Dyuktai Goose (Anser djuktaiensis) and Hooded Crane (Grus monacha) from the Pleistocene of central Yakutia. Biological Sciences 499: 99-102. 2021
[5] Hiroshige Matsuoka; Yoshikazu Hasegawa: Annakacygna, a new genus for two remarkable flightless swans (Aves, Anatidae, Cygnini) from the Miocene of Gunna, central Japan: with a note on the birds’ food niche shift and specialization of wings for parental care. Bulletin of Gunma Museum of Natural History 26: 1-30. 2022
[6] Trevor H. Worthy; R. Paul Scofield; Suzanne J. Hand; Vanessa L. De Pietri; Michael Archer: A swan-sized fossil anatid (Aves: Anatidae) from the early Miocene St Bathans Fauna of New Zealand. Zootaxa 5168(1): 39-50. 2022
[7] Gerald Mayr; Thomas Lechner; Madelaine Böhme: Nearly complete leg of an unusual shelduck-sized Anseriformes bird from the earliest late Miocene hominid locality Hammerschmiede (Germany). Historical Biology, DOI: 10.1080/08912963.2022.2045285. 2022
*********************
bearbeitet: 14.02.2024
Fossile Cathartiformes
Cathartidae
Aizenogyps toomeyae Emslie
Brasilogyps faustoi Alvarenga
Breagyps clarki Miller
Cathartes emsliei Suárez & Olson [2]
Coragyps occidentalis Miller
Coragyps seductus Suárez [3]
Diatropornis ellioti (Milne-Edwards)
Dryornis hatcheri Degrange [5]
Dryornis pampeanus Moreno & Mercerat [4]
Geronogyps reliquus Campbell
Gymnogyps amplus Miller
Gymnogyps howardae Campbell
Gymnogyps kofordi Emslie
Gymnogyps varonai (Arredondo)
Hadrogyps aigialeus Emslie
Pampagyps imperator Agnolin et al. [1]
Perugyps diazi Stucchi & Emslie
Phasmagyps patritus Wetmore
Pleistovultur nevesi Alvarenga et al.
Pliogyps charon Becker
Pliogyps fisheri Tordoff
Sarcoramphus fisheri Campbell
Vultur messii Degrange et al.
Wingegyps cartellei Alvarenga & Olson
Teratornithidae
Aiolornis incredibilis (Howard)
Argentavis magnificens Campbell & Tonni
Cathartornis gracilis Miller
Oscaravis olsoni (Arredondo & Arredondo)
Taubatornis campbelli Olson & Alvarenga
Teratornis merriami Millner
Teratornis woodburnensis Campbell & Stenger
*********************
Quellen:
[1] Federico L. Agnolin; Federico Brissón Egli; Esteban Soibelzon; Sergio G. Rodriguez; Leopoldo H. Soibelzon; Facundo Iacona; David Piazza: A new large Cathartidae from the Quaternary of Argentina, with a review of the fossil record of condors in South America. Contribuciones Científicas del Museo Argentino de Ciencias Naturales “Bernardino Rivadavia” 7: 1-16. 2017
[2] William Suárez; Storrs L. Olson: A new fossil vulture (Cathartidae: Cathartes) from Quaternary asphalt and cave deposits in Cuba. Bulletin of the British Ornithologists’ Club 140(3): 335-343. 2020
[3] William Suárez: The fossil avifauna of the tar seeps Las Breas de San Felipe, Matanzas, Cuba. Zootaxa 4780(1). 2020
[4] Federico J. Degrange; Claudia P. Tambussi; Matías L. Taglioretti; Fernando A. Scaglia: Phylogenetic affinities and morphology of the Pilocene cathartiform Dryornis pampeanus Moreno & Mercerat. Papers in Palaeontology 2021
[5] F. J. Degrange: A new species of Dryornis (Aves, Cathartiformes) from the Santa Cruz Formation (lower Miocene), Patagonia, Argentina. Journal of Vertebrate Paleontology advance online publication. doi: 10.1080/02724634.2021.2008411. 2022
*********************
bearbeitet: 19.12.2023
Fossile Accipitriformes
Family incertae sedis
Aviraptor longicrus Mayr & Hurum [3]
Accipitridae
Anchigyps voorhiesi Zhang, Feduccia & James
Apatosagittarius terrenus Feduccia & Voorhies
Archaehierax sylvestris Mather, Lee, Camens & Worthy [6]
Buteo dundasi Degrange et al. [5]
Buteo sanfelipensis Suárez [3]
Buteogallus royi Suárez [4]
Cryptogyps lacertosus (de Vis) [7]
Dynatoaetus gaffae Mather et al. [9]
Dynatoaetus pachyosteus Mather et al.
Garganoaetus freudenthali Ballmann
Garganoaetus murivorus Ballmann
Gigantohierax itchei Suárez [4]
Gypaetus georgii Sánchez-Marco [8]
Gyps bochenskii Boev
Gyps melitensis Lydekker
Neogyps errans Miller
Neophron lolis Sánchez-Marco [8]
Neophrontops americanus Miller
Neophrontops slaughteri Feduccia
Neophrontops vallecitoensis Howard
Neophrontops vetustus Wetmore
Palaeoplancus dammanni Mayr & Perner [1]
Vinchinavis paka Tambussi et al. [2]
Horusornithidae
Horusornis vianeyliaudae Mourer-Chauviré
Pandionidae
Pandion homalopteron Warter
Pandion lovensis Becker
Pandion pannonicus Kessler
Pandionidae gen. & sp. ‚Bad Münster am Stein, Deutschland‘
Sagittariidae
Amanuensis pickfordi Mourer-Chauviré
Pelargopappus magnus Milne-Edwards
*********************
References:
[1] Gerald Mayr; Thomas Perner: A new species of diurnal birds of prey from the late Eocene of Wyoming (USA) – one of the earliest New World records of the Accipitridae (hawks, eagles, and allies). Neues Jahrbuch für Geologie und Paläontologie – Abhandlungen 297(2): 205 – 215. 2020
[2] Claudia P. Tambussi; Federico J. Degrange; Patricia L. Ciccioli; Francisco Prevosti: Avian remains from the Toro Negro Formation (Neogene), Central Andes of Argentina. Journa of South American Earth Sciences 2020
[3] Gerald Mayr; Jørn H. Hurum: A tiny, long-legged raptor from the early Oligocene of Poland may be the earliest bird-eating diurnal bird of prey. The Science of Nature 107(48): 2020. https://doi.org/10.1007/s00114-020-01703-z
[4] William Suárez: The fossil avifauna of the tar seeps Las Breas de San Felipe, Matanzas, Cuba. Zootaxa 4780(1). 2020
[5] Federico J. Degrange, Claudia P. Tambussi, Matías L. Taglioretti, and Fernando A. Scaglia: A new buzzard from the late Pliocene of Argentina. Acta Palaeontologica Polonica. https://doi.org/10.4202/app.00933.2021
[6] Ellen K. Mather; Michael S. Y. Lee; Aaron B. Camens; Trevor H. Worthy: An exceptional partial skeleton of a new basal raptor (Aves: Accipitridae) from the late Oligocene Namba formation, South Australia. Historical Biology – An International Journal of Paleobiology, September 2021
[7] E. K. Mather; M. S. Lee; T. H. Worthy: A new look at an old Australian raptor places “Taphaetus” lacertosus de Vis 1905 in the Old World vultures (Accipitridae: Aegypiinae). Zootaxa 1–23. 2022
[8] Antonio Sánchez-Marco: Two new Gypaetinae (Accipitridae, Aves) from the late Miocene of Spain. Historical Biology. https://doi.org/10.1080/08912963.2022.2053117. 2022
[9] Ellen K. Mather; Michael S. Y. Lee; Aaron B. Camens; Trevor S. Worthy: A giant raptor (Aves: Accipitridae) from the Pleistocene of southern Australia. Journal of Ornithology. https://doi.org/10.1007/s10336-023-02055-x. 2023
*********************
bearbeitet: 22.11.2023
Fossile Procellariiformes
Familie incertae sedis
Makahala mirae Mayr
Diomedeidae
Aldiomedes angustirostris Mayr & Tennyson
Diomedea milleri Howard
Diomedea thyridata Wilkinson
Diomedavus knapptonensis Mayr & Goedert
Murunkus subitus Panteleyev & Nessov (?)
Notoleptos giglii Acosta Hospitaleche & Gelfo
Phoebastria anglica Lydekker
Phoebastria immutabilis Rothschild
Phoebastria rexularum Olson & Rasmussen
Plotornis archaeonautes Ksepka et al.
Plotornis arvernensis (Milne-Edwards in Shufeldt)
Plotornis delfortrii Milne-Edwards
Plotornis graculoides Portis
Tydea septentrionalis Mayr & Smith
Diomedeoididae
Diomedeoides babaheydariensis Peters & Hamedani
Diomedeoides brodkorbi Cheneval
Rupelornis definitus van Beneden
Hydrobatidae
Oceanodroma hubbsi Miller
Pelecanoididae
Pelecanoides cymatotrypetes Olson
Pelecanoides miokuaka Worthy et al.
Procellariidae
Ardenna buchananbrowni Tennyson et al.
Ardenna conradi Marsh
Ardenna davealleni Tennyson & Mannering
Ardenna gilmorei Chandler
Argyrodyptes microtarsus Ameghino
Calonectris krantzi Olson & Rasmussen
Calonectris kurodai Olson
Calonectris wingatei Olson
Eopuffinus kazachstanensis Nessov
Fulmarus hammeri Howard
Fulmarus miocaenus Howard
Hydrornis natator Milne-Edwards
Macronectes tinae Tennyson & Salvador
Oestrelata vociferans Shufeldt
Pachyptila salax Olson
Procellaria altrirostris Tennyson & Tomotani [1]
Procellaria antiqua Milne-Edwards
Pterodroma kurodai Harrison & Walker
Pterodromoides minoricensis Segui et al.
Puffinus barnesi Howard
Puffinus calhouni Howard
Puffinus diatomicus Miller
Puffinus eyermani Shufeldt
Puffinus felthami Howard
Puffinus inceptor Wetmore
Puffinus kanakoffi Howard
Puffinus micraulax Brodkorb
Puffinus mitchelli Miller
Puffinus nestori Alcover
Puffinus parvus Shufeldt
Puffinus priscus Miller
Puffinus raemdonckii (van Beneden)
Puffinus sp. ‘Lee Creek Mine, USA 1’
Puffinus sp. ‘Lee Creek Mine, USA 2’
Puffinus tedfordi Howard
Tytthostonychidae
Tytthostonyx glauconiticus Olson & Parris
*********************
Referenzen:
[1] A. J. D. Tennyson; B. M. Tomotani: A new fossil species of Procellaria (Aves: Procellariiformes) from tzhe Pliocene of New Zealand. Papéis Avulsos De Zoologia, 61, e20216116. 2021
*********************
bearbeitet: 09.04.2024
Aus eins mach drei – Schwarzflügelmonarch
Der Schwarzflügelmonarch (Monarcha frater Sclater) ist ein typischer Vertreter der Familie Monarchidae und lebt in Australien und Neuguinea, es werden vier Unterarten unterschieden.
Die Art wird nun nach einer Studie aus dem Jahr 2023 in drei monotypische Arten aufgespalten (zumindest wird eine solche Aufspaltung vorgeschlagen).:
Perlmonarch (Monarcha canescens Salvadori)
Arfak-Monarch (Monarcha frater Sclater)
Maskenmonarch (Monarcha periophthalmicus Sharpe) (ssp. kunupi = synonym)
*********************

Foto: Tony Palliser
https://www.inaturalist.org/people/tonypalliser
https://creativecommons.org/licenses/by-nc/4.0/

Foto: David Bishop
https://www.inaturalist.org/people/kdbishop69
https://creativecommons.org/licenses/by-nc/4.0/
*********************
Quelle:
[1] Leo Joseph; Julian Teh; Paul Sweet; Phil Gregory: The Black-winged Monarch (Monarcha frater): Geographic variation, taxonomy, a “new” population, and an enduring mystery in migration. Avian Research 14(3): 1-11. 2023
*********************
bearbeitet: 08.11.2023
Azurracke – eine eigenständige Art oder nicht?
Azurracke (Eurystomus azureus G. R. Gray)
Die Azurracke ist auf die nördlichen Molukken, Indonesien beschränkt, sie weist ein nahezu einheitliches azurblaues Gefieder auf und unterscheidet sich dadurch deutliche von den neun derzeit anerkannten Unterarten der Türkisracke (Eurystomus orientalis (L.)), die auch sehr viel weiter verbreitet ist.
Eine DNA-Studie aus dem Jahr 2018 kam zu dem Ergebnis, dass die Azurracke innerhalb der Unterarten der Türkisracke anzusiedeln ist, sprich, dass sie genetisch gesehen eine Unterart der Türkisracke darstellt.
Eine weitere Studie aus dem Jahr 2023 bestätigt nun dieses Ergebnis, schlägt aber vor die verschiedenen Formen der Türkisracke weiter zu untersuchen, denn wenn man der Azurracke weiterhin den Status einer eigenen Art zuerkennen will, so muss man die genetisch deutlich gekennzeichneten Unterartengruppen der Türkisracke ebenfalls als jeweils eigenständige Arten anerkennen.:
Unterschieden werden können eine nördliche Gruppe (Klade), eine südliche- sowie eine östliche Gruppe; da sich azureus aber innerhalb eben dieser letztgenannten östlichen Gruppe findet, müsste diese weiter aufgesplittet werden, dies ergäbe dann eine Australasiatische Gruppe und eine Gruppe, die auf die Salomonen beschränkt ist – im Ganzen wären dies nun also vier neue Arten.
Die neuen Arten würden nun heißen:
Nördliche Türkisracke (Eurystomus cyanicollis Vieillot)
Türkisracke (Eurystomus orientalis (L.))
Australische Türkisracke (Eurystomus pacificus (Latham))
Salomonen-Türkisracke (Eurystomus solomonensis Sharpe)
*********************

Foto aus: ‘Ulf S. Johansson; Martin Irestedt; Per G. P. Ericson: Patterns of phylogenetic diversification in the Dollarbird (Eurystomus orientalis) and Azure Roller (Eurystomus azureus) complex. Molecular Phylogenetics and Evolution 189: 1-13. 2023’
https://creativecommons.org/licenses/by-nc/4.0/
*********************
Quellen:
[1] Ulf S. Johansson; Martin Irestedt; Yanhua Qu; Per G. P. Ericson: Phylogenetic relationships of rollers (Coraciidae) based on complete mitochondrial genomes and fifteen nuclear genes. Molecular Phylogenetics and Evolution 126: 17-22. 2018
[2] Ulf S. Johansson; Martin Irestedt; Per G. P. Ericson: Patterns of phylogenetic diversification in the Dollarbird (Eurystomus orientalis) and Azure Roller (Eurystomus azureus) complex. Molecular Phylogenetics and Evolution 189: 1-13. 2023
*********************
bearbeitet: 08.11.2023
Through Time And Clades – Dinosaurs, the Second Chapter
Part 1:
Part 2:
Part 3:
Part 4:
Part 5:
Part 6:
… there’s more to come
Part 7:
Part 8:
Part 9:
Part 10:
Part 11:
Part 12:
Part 13:
Part 14:
Part 15.1:
Part 15.2:
Updates:
more Updates:
*********************
edited: 03.11.2023
The Largest Avian Radiation: The Evolution of Perching Birds, or the Order Passeriformes
Jon Fjeldså, Les Christidis, Per G. P. Ericson: The Largest Avian Radiation: The Evolution of Perching Birds, or the Order Passeriformes. Lynx 2020
*********************

I think I first heard about this book sometimes on the notorious blue-bird-pages aka. shitter, no twitter ….
Anyways, this is easily one of the best books I have ever read; as its title already suggests, it deals with the greatest radiation in the bird kingdom – the Passeriformes.
Every family, and every new family is mentioned and described, some future splits are predicted (subfamilies in the book, that now, three years later, are accepted as full families); it is just breathtaking, and I learned so much new stuff!
Every family is depicted with at least one, sometimes more representative species, all painted by Jon Fjeldså himself; in some cases I even know the original photos he used for his depictions.
This is a book that I simply have to recommend for every bird enthusiast!
*********************
edited: 02.11.2023
Blue Tit

*********************
edited: 18.10.2023
Coal Tit

*********************
edited: 18.10.2023
European Jackdaw

*********************
edited: 18.10.2023
Fossil record of the Pterocliformes
Pteroclidae
Linxiavis inaquosus Li, Stidham, Deng & Zhou
Pterocles bosporanus Zelenkov
Pterocles larvatus Milne-Edwards
Pterocles sepultus Milne-Edwards
Pterocles validus Milne-Edwards
*********************
References:
[1] Zhiheng Li; Thomas A. Stidham; Tao Deng; Zhonghe Zhou: Evidence of Late Miocene Peri-Tibetan aridification from the oldest Asian species of sandgrouse (Aves: Pteroclidae). Frontiers in Ecology and Evolution, 31 March 2020
*********************
edited: 16.10.2023
Birds unknown – the Spotted Wren-Babbler
Spotted Wren-Babbler (Elachura formosa Walden)
***
The Spotted Wren-Babbler or Spotted Elachura (or just Elachura) is an approximately 10 cm long, inconspicuous bird that inhabits the understory of dense forests in South- and Southeast Asia.
The species was originally described as a wren, later assigned to the timalia family and finally placed in its own monotypic family because it does not appear to be related to any other bird species; however, it has similarly shaped foot soles as the waxwings (Bombycilidae) and goldcrests (Regulidae), which are also taxonomically quite isolated, but no further studies have been carried out on this yet. [1][2]
*********************

https://www.inaturalist.org/people/udayagashe
https://creativecommons.org/licenses/by-nc/4.0/
*********************
References:
[1] Per Alström; Daniel M. Hooper; Yang Liu; Urban Olsson; Dhananjai Mohan; Magnus Gelang; Hung Le Manh; Jian Zhao; Fumin Lei; Trevor D. Price: Discovery of a relict lineage and monotypic family of passerine birds. Biology Letters 10(3): 1-5. 2014
[2] Jon Fjeldså; Les Christidis; Per G. P. Ericson: The Largest Avian Radiation: The Evolution of Perching Birds, or the Order Passeriformes. Lynx Edicions 2020
*********************
edited: 09.10.2023
Birds unknown – the Grande Comore Blue Vanga
Grande Comore Blue Vanga (Cyanolanius comorensis ssp. bensoni Louette & Herremans)
***
This taxon, described in 1982, is thought to inhabit, or to have inhabited, the island of Grande Comore, Comoros in the Indian Ocean; it is, however, known from only a single somewhat doubtful specimen that was collected as late as 1981.
Here is an excerpt from the original description.:
“In bill length intermediate between C. m. madagascarinus and comorensis; total culmen 18.5 mm as against an average of 17.3 in both sexes of madagascarinus and 20.1 mm in both sexes of comorensis. Bill decidedly more robust than in madagascarinus, although not much longer. In plumage in all respects seemingly nearest to comorensis although there is no similar aged specimen of this race available.“
“Apart from the diagnosis of bensoni, the following is a fuller description of the holotype (not fully adult):- Upperparts lilac blue, crown somewhat darker, with a few bright blue (adult) feathers appearing on crown and mantle. Inner webs of inner secondaries blue, like outer webs (like comorensis, inner webs not black as in madagascarinus). Underparts white, a few buffish feathers on flanks. Black mask just starting to appear. Tail feathers with square tips, not pointed as in very young specimens of Cyanolanius, and with buffish fringes. Iris pale blue. Legs grey-blue. Bill with pale base to both mandibles, tips dark. One may conclude that bensoni agrees well with comorensis in plumage characteristics but has a definitely less robust bill, intermediate in size between the 2 other races. Some doubt may persist as to the bill size in the adult, but I have measurements from 4 immatures of madagascarinus (mounted) in RNL, certainly somewhat younger than the holotype of bensoni, averaging 17.1 (versus 17.3 in the adult, see above) showing that this possible difference is insignificant.”
***
This sole specimen may represent a stray from the nearby island of Mohéli, which is home to the nominate race of the Comoros Blue Vanga (Cyanolanius comorensis (Shelley)), or it may indeed constitute a member of a distinct endemic population. If there indeed ever was a population of Blue Vangas living on Grande Comore, it must now be extinct.
There is also another mystery – why are there no Blue Vangas to be found (at least today) on the islands of Anjouan and Mayotte which in fact lie between Mohéli and Madagascar?
*********************

Photo: RMCA, Tervuren
https://creativecommons.org/licenses/by-nc-sa/3.0/
*********************
References:
[1] M. Louette; M. Herremans: The Blue Vanga Cyanolanius madagascarius on Grand Comoro. Bulletin of the British Ornithologists’ Club 102(4): 132-135.1982
*********************
edited: 04.10.2023
From seven to thirteen – the genus Schiffornis
The species of the genus Schiffornis inhabit the tropics of northern South America, especially the Amazon region; up to now this genus was thought to contain seven species with several subspecies, however, splitting had already been suggested for some time.:
Foothill Schiffornis (Schiffornis aenea (Zimmer))
Varzea Schiffornis (Schiffornis major Des Murs)
Guianan Schiffornis (Schiffornis olivacea (Ridgway))
Russet-winged Schiffornis (Schiffornis stenorhyncha (Sclater, PL & Salvin))
Brown-winged Schiffornis (Schiffornis turdina (Wied))
Northern Schiffornis (Schiffornis veraepacis (Sclater, PL & Salvin))
Greenish Schiffornis (Schiffornis virescens (Lafresnaye))
Now, a new study found that the diversity indeed is much higher than previously thought, and that this genus actually contains many more, yet very cryptic, species.
One was described as completely new.:
Cracraft’s Schiffornis (Schiffornis cracrafti Lima et al.)
The others have been upleveled from subspecific to specific rank (Unfortunately I cannot properly name them here because I cannot access the paper discussed here.).
Most of these new species cannot really be distinguished by their appearance but differ significantly by their songs and by their DNA. [1]
*********************

Photo: edvandroabreuribeiro
https://www.inaturalist.org/people/edvandroabreuribeiro
https://creativecommons.org/licenses/by-nc/4.0/
*********************
References:
[1] Rafael Dantas Lima; Fernanda Bocalini; Luís Fábio Silveira: Integrative revision of species limits in the genus Schiffornis (Aves: Tityridae) reveals cryptic diversity in the Neotropics. Zoological Journal of the Linnean Society advance online publication. doi: 10.1093/zoolinnean/zlad108. 2023
*********************
edited: 04.10.2023
Fossile Arten der Charadriiformes
Familie incertae sedis
Charadriisimilis essexensis Mayr & Kitchener
Cherevychnavis umanskae Bochenski, Wertz, Tomek, & Gorobets
Chionoides australiensis De Pietri et al.
Elorius limosoides De Pietri & Mayr
Elorius paludicola Milne-Edwards
Hakawai melvillei De Pietri, Scofiled, Tennyson, Hand & Worthy
Jiliniornis huadianensis Hou & Ericson
Nahmavis grandei Musser & Clarke [1]
Neilus sansomae De Pietri et al.
Palintropus retusus (Marsh)
Parvelorius calidris De Pietri & Mayr
Parvelorius gracilis (Milne-Edwards)
Scandiavis mikkelseni Bertelli, Lindow, Dyke & Mayr
Scolopacimilis lartetianus (Milne-Edwards)
Scolopacimilis sp. ‚Saint-Gerand-le-Puy, Frankreich‘
Sternalara milneedwardsi De Pietri et al.
Sternalara minuta De Pietri et al.
Vanolimicola longihallucis Mayr
Alcidae/Pan-Alcidae
Aethia barnesi Smith
Aethia rossmoori Howard
Aethia storei Smith
Alca ausonia Portis
Alca carolinensis Smith & Clarke
Alca grandis Brodkorb
Alca minor Smith & Clarke
Alca olsoni Smith & Clarke
Alca stewarti Martin et al.
Alcodes ulnulus Howard
Brachyramphus dunkeli Chandler
Brachyramphus pliocenum Howard
Cepphus olsoni Howard
Cerorhinca dubia Miller
Cerorhinca minor Howard
Cerorhinca reai Chandler
Divisulcus demerei Smith
Fratercula dowi Guthrie et al.
Hydrotherikornis oregonus Miller
Mancalla californiensis Lucas
Mancalla cedrosensis Howard
“Mancalla” diegensis Miller
“Mancalla” emlongi Olson
Mancalla lucasi Smith
“Mancalla” milleri Howard
Mancalla vegrandis Smith
Miocepphus blowi Wijnker & Olson
Miocepphus bohaskai Wijnker & Olson
Miocepphus mcclungi Wetmore
Miocepphus mergulellus Wijnker & Olson
Miomancalla howardi (Smith)
Miomancalla wetmorei (Howard)
Praemancalla lagunensis Howard
Pseudocepphus teres Wijnker & Olson
Synthliboramphus rineyi Chandler
Uria affinis Marsh
Uria brodkorbi Howard
Uria paleohesperis Howard
Uria troile Lydekker
Burhinidae
Burhinus lucorum Bickart
Genucrassum bransatensis De Pietri & Scofield
Charadriidae
Charadrius lambrechti Kessler
Vanellus liffyae De Pietri, Scofield, Prideaux & Worthy
Cimolopterygidae (?)
Ceramornis major Brodkorb
Cimolopteryx maxima Brodkorb
Cimolopteryx rara Marsh
Lamarqueavis australis Agnolin
Lamarqueavis minima (Brodkorb)
Lamarqueavis petra (Hope)
Glareolidae
Becassius charadriioides De Pietri, Mayr & Scofield
Boutersemia belgica Mayr & Smith
Boutersemia parvula Mayr & Smith
“Gallinago” veterior Jánossy
Glareola neogena Ballmann
Mioglareola dolnicensis (Švec)
Mioglareola gregaria Ballmann
Paractiornis perpusillus Wetmore
Pinguinus alfrednewtoni Olson
Graculavidae (?)
Dakotornis cooperi Erickson
Graculavus augustus Hope
Graculavus velox (Marsh)
Palaeotringa littoralis Marsh
Palaeotringa vagans Marsh
Scaniornis lundgreni Dames
Telmatornis priscus Marsh
Zhylgaia aestiflua Nesov
Haematopodidae
Haematopus sulcatus (Brodkorb)
Jacanidae
Jacana farrandi Olson
Janipes nymphaeobates Rasmussen et al.
Nupharanassa bulotorum Rasmussen et al.
Nupharanassa tolutaria Rasmussen et al.
Laornithidae (?)
Laornis edvardsianus Marsh
Laricolidae
Laricola desnoyersii (Milne-Edwards)
Laricola elegans (Milne-Edwards)
Laricola intermedia De Pietri et al.
Laricola robusta De Pietri et al.
Laricola totanoides (Milne-Edwards)
Laridae
Larus dolnicensis Švec
Larus elmorei Brodkorb
Larus lacus Emslie
Larus perpetuus Emslie
Larus pristinus Shufeldt
Larus oregonus Shufeldt
Larus robustus Shufeldt
Pedionomidae
Oligonomus milleri De Pietri, Camens & Worthy
Recurvirostridae
Fluviatilavis antunesi Harrison
Himantopus brevipes Milne-Edwards
Recurvirostra teruelensis (Villalta) [2]
Rostratulidae
Rostratula minator Olson & Eller
Scolopacidae
Bartramia umatilla Brodkorb
Calidris binagadensis Serebrovsky
Calidris janossyi Kessler
Calidris pacis Brodkorb
Erolia penepusilla Brodkorb
Ereunetes rayi Brodkorb
Gallinago veterior Jánossy
Micropalama chapmani Campbell
Micropalama hesternus Wetmore
Scolopax baranensis Jánossy
Scolopax carmesinae Seguí
Stercorariidae
Stercorarius shufeldti Howard
Thinocoridae
Thinocorus koepckae Campbell
Turnicidae
Cerestenia pulchrapenna Mayr
Turnipax dissipata Mayr
Turnipax oechslerorum Mayr & Knopf
*********************
Quellen:
[1] Grace Musser; Julia A. Clarke: An exceptionally preserved specimen from the Green River Formation elucidates complex phenotypic evolution in Gruiformes and Charadriiformes. Frontiers in Ecology and Evolution. 8:559929. doi: 10.3389/fevo.2020.559929. 2020
[2] Antonio Sánchez-Marco: Old and new fossil birds from the Spanish Miocene. Journal of Iberian Geology 47(4): 697-712. 2021
*********************
bearbeitet: 29.09.2023
Fossil record of the Strigiformes
Family incertae sedis
Enskenia galeanoi Agnolín
Primoptynx poliotauros Mayr, Gingerich & Smith [5]
Ypresiglaux michaeldanielsi Mayr & Kitchener [8]
Heterostrigidae
Heterostrix tatsinensis Kurochkin & Dyke
Ogygoptyngidae
Ogygoptynx wetmorei Rich & Bohaska
Palaeoglaucidae
Palaeoglaux artophoron Peters
Palaeoglaux perrierensis Mourer-Chauviré
Protostrigidae
Eostrix gulottai Mayr
Eostrix martinellii Wetmore
Eostrix mimica (Wetmore)
Eostrix tsaganica Kurochkin & Dyke
“Eostrix” vincenti Harrison
Minerva antiqua (Shufeldt)
Minerva californiensis (Howard)
Minerva leptosteus (Marsh)
Minerva lydekkeri Shufeldt
Minerva saurodosis Wetmore
Oligostrix rupeliensis Fischer
Sophiornithidae
Berruornis halbedeli Mayr
Berruornis orbisantiqui Mourer-Chauviré
Palaeobyas cracrafti Mourer-Chauviré
Palaeotyto cadurcensis Mouer-Chauviré
Sophiornis quercynus Mourer-Chauviré
Strigidae
Aegolius funereus (L.)
Alasio collongensis (Ballmann)
Asio brevipes Ford & Murray
Asio ecuadoriensis Lo Coco, Agnolín & Carrión [2]
“Asio” henrici Milne-Edwards
Asio longaevus (Umanskaya)
Asio priscus Howard
Athene angelis Mourer-Chauviré
Athene cretensis Weesie
Athene cunicularia ssp. intermedia Feduccia
Athene megalopeza (Ford)
Athene noctua ssp. lunellensis Mourer-Chauviré
Athene noctua ssp. veta Jánossy
Athene trinacriae Pavia & Mourer-Chauviré
Athene vallgornerensis Guerra, Bover & Alcover [1]
Bubo binagadensis Burchak-Abramovich
Bubo bubo ssp. davidi Mourer-Chauviré
Bubo (?) florianae Kretzoi
Bubo ibericus Meijer, Pavia, Madurell-Malapeira & Alba
Bubo leakeyae Brodkorb & Mourer-Chauviré
Bubo lignitum Giebel
Bubo osvaldoi Arrendondo & Olson
Bubo perpasta Ballmann
Bubo scandiacus ssp. gallicus (Mourer-Chauviré)
Bubo sp. ‘Soave Cava Sud, Italy’
Bubo zeylonensis ssp. lamarmorae Mlíkovský
Glaucidium explorator Emslie
Glaucidium ireneae Pavia [3]
Intulula brevis (Ballmann)
Intulula tinnipara Mlíkovský
Margarobyas abronensis Zelenkov & Gonzáles [7]
Mioglaux debellatrix Mlíkovský
Mioglaux poirrieri (Milne-Edwards)
Miosurnia diurna Li et al. [9]
Ornimegalonyx ewingi Suárez [4]
Ornimegalonyx gigas Arredondo
Otus guildayi Brodkorb & Mourer-Chauviré
Otus wintershofensis (Ballmann)
Pulsatrix arredondoi Brodkorb
Strix brea Howard
“Strix” brevis Ballmann
Strix collongensis Ballman
Strix dakota A. H. Miller
Strix edwardsi (Ennouchi)
Strix intermedia Jánossy
Strix sp. ‘Ladds, USA’
Surnia capeki Jánossy
Surnia robusta Jánossy
Yarquen dolgopolae Tambussi et al.
Tytonidae
Basityto rummeli Mlíkovský
Miotyto montispetrosi Göhlich & Ballmann
Necrobyas arvernensis (Milne-Edwards)
Necrobyas harpax Milne-Edwards
Nocturnavis incerta (Milne-Edwards)
Prosybris antiqua (Milne-Edwards)
Prosybris media (Mourer-Chauviré)
Selenornis henrici (Milne-Edwards)
Tyto balearica (Mourer-Chauviré)
Tyto balearica ssp. cyrneichnusae Louchart
Tyto campiterrae Jánossy
Tyto gigantea Ballmann
Tyto jinniushanensis Hou
Tyto maniola Súarez & Olson [6]
Tyto mourerchauvireae Pavia
Tyto robusta Ballmann
Tyto sanctialbani (Lydekker)
*********************
References:
[1] Carmen Guerra; Pere Bover; Josep Antoni Alcover: A new species of extinct little owl from the Pleistocene of Mallorca (Balearic Islands). Journal of Ornithology 153(2): 347-354. 2012
[2] Gastón E. Lo Coco; Federico L. Agnolín; José Luis Román Carrión: Late Pleistocene owls (Aves, Strigiformes) from Ecuador, with the description of a new species. Journal of Ornithology 161: 713-721. 2020
[3] Marco Pavia: Palaeoenvironmental reconstruction of the Cradle of Humankind during the Plio-Pleistocene transition, inferred from the analysis of fossil birds from Member 2 of the hominin-bearing site of Kromdraai (Gauteng, South Africa) Quaternary Science Reviews. 248: Article 106532. doi:10.1016/j.quascirev.2020.106532. 2020
[4] William Suárez: Remarks on extinct giant owls (Strigidae) from Cuba, with description of a new species of Ornimegalonyx Arrendondo. Bulletin of the British Ornithologists’ Club 140(4): 387-392. 2020
[5] Gerald Mayr; Philip D. Gingerich; Thierry Smith: Skeleton of a new owl from the early Eocene of North America (Aves, Strigiformes) with an accipitrid-like foot morphology. Journal of Vertebrate Paleontology. doi: 10.1080/ 02724634.2020.1769116. 2020
[6] William Súarez; Storrs L. Olson: Systematics and distribution of the living and fossil small barn owls of the West Indies (Aves: Strigiformes: Tytonidae). Zootaxa 4830(3): 544-564. 2020
[7] Nikita V. Zelenkov; Soraida F. Gonzáles: A new extinct species of Margarobyas (Strigiformes) and the evolutionary history of the endemic Cuban bare-legged owl (M. lawrencii). Journal of Vertebrate Paleontology. https://doi.org/10.1080/02724634.2021.1995869. 2021
[8] G. Mayr; A. C. Kitchener: Early Eocene fossil illuminates the ancestral (diurnal) ecomorphology of owls and documents a mosaic evolution of the strigiform body plan. Ibis. doi: 10.1111/ibi.13125. 2022
[9] Z. Li, T. A. Stidham; X. Zheng; Y. Wang; T. Zhao; T. Deng; Z. Zhou: Early evolution of diurnal habits in owls (Aves, Strigiformes) documented by a new and exquisitely preserved Miocene owl fossil from China. PNAS 119: e2119217119. doi: 10.1073/pnas.2119217119. 2022
*********************
edited: 18.09.2023
Fossile Coraciiformes
Familie incertae sedis
Chehuenia facongrandei Agnolín
Microena goodwini Harrison & Walker
Microena sp. ‘Isle of Sheppey’
Paracoracias occidentalis Clarke et al.
Quasisyndactylus longibrachis Mayr
Alcedinidae
Halcyoninae gen. & sp. ‘Riversleigh, Australien’
Coraciidae
Eurystomus beremendensis Kessler
Miocoracias chenevali Mourer-Chauviré, Peyrouse & Hugueney
Eocoraciidae
Eocoracias brachyptera Mayr & Mourer-Chauviré
Geranopteridae
Geranopterus alatus Milne-Edwards
Geranopterus bohemicus Mlikovský
Geranopterus milneedwardsi Mayr & Mourer-Chauviré
Momotidae
Momotidae gen. & sp. ‘Florida, USA’
Protornis glarniensis von Meyer (?)
Primobucconidae
Primobucco frugilegus Mayr & Mourer-Chauviré
Primobucco mcgrewi Brodkorb
Primobucco perneri Mayr & Mourer-Chauviré
Septencoracias morsensis Bourdon et al.
Septencoracias simillimus Mayr & Kitchener
Todidae
Palaeotodus emryi Olson
Palaeotodus escampsiensis Mourer-Chauviré
Palaeotodus itardiensis Mourer-Chauviré
Todus sp. ‘MNHN 73.5395’ [2]
***
Merops radobojensis (Meyer) wurde offenbar anhand eines Bruchstücks einer Elle beschrieben, das tatsächlich nicht dieser Gattung zugeordnet werden kann, sondern eher von einem Sperlingsvogel zu stammen scheint.
Daher habe ich den Namen entfernt. [1]
*********************
Quellen:
[1] Nikita V. Zelenkov: The revised avian fauna of Rudabànya (Hungary, Late Miocene). In: Paleontología y evolución de las Aves. Eds. C. Acosta Hospitaleche, F. L. Agnolin, N. Haidr, J. I. Noriega, C. P. Tambussi. Contribuciones del Museo Argentino de Ciencias Naturales 7: 253-266. 2017
[2] N. V. Zelenkov; S. F. Gonzalez: The first fossil Tody (Aves: Todidae) from Cuba. Paleontological Journal 54(4): 414-419. 2020
[3] Federico J. Degrange; Diego Pol; Pablo Puerta; Peter Wilf: Unexpected larger distribution of paleogene stem-rollers (AVES, CORACII): new evidence from the Eocene of Patagonia, Argentina. Scientific Reports 11: 1363. 2021
*********************
bearbeitet: 16.04.2024
Fossil record of the Tinamiformes
Tinamidae
Crypturellus reai Chandler
Eudromia intermedia (Rovereto)
Eudromia olsoni Tambussi & Tonni
Mininothura talenki Agnolín
Nothura paludosa Mercerat
Nothura parvula (Rovereto)
Querandiornis romani Rusconi
*********************
edited: 18.09.2023
Fossil record of the Falconiformes
Falconidae
Asturaetus furcillatus De Vis
Badiostes patagonicus Ameghino
Baza gracilis De Vis
Caracara creightoni Brodkorb
Caracara latebrosus Brodkorb
Caracara major Jones, Rinderknecht, Migotto & Blanco
Caracara seymouri Suárez & Olson
Caracara sp. ‘Río Quequén Grande, Argentinia’
Caroohierax rapoporti Agnolín
Falco pisanus Portis
Falco powelli Emslie & Mead
Falco ramenta Wetmore
Foetopterus ambiguus Moreno & Mercerat
Lagopterus minutus Moreno & Mercerat
Milvago brodkorbi Campbell
Milvago diazfrancoi Suárez
Polyborus latebrosus Wetmore
Sushkinia pliocaenica Tugarinov
Taphaetus brachialis De Vis
Taphaetus lacertosus De Vis
Thegornis debilis Ameghino
Thegornis musculosus Ameghino
Thegornis sosae Agnolín
Thegornis spivacowi Agnolín
Masillaraptoridae (?)
Danielsraptor phorusrhacoides Mayr & Kitchener [2]
Masillaraptor parvunguis Mayr
*********************
References:
[1] William Suárez: The fossil avifauna of the tar seeps Las Breas de San Felipe, Matanzas, Cuba. Zootaxa 4780(1). 2020
[2] Gerald Mayr; Andrew C. Kitchener: New fossils from the London Clay show that the Eocene Masillaraptoridae are stem group representatives of falcons (Aves, Falconiformes). Journal of Vertebrate Paleontology. 2022
*********************
edited:18.09.2023
Fossile Gruiformes
Familie incertae sedis
Bumbanipes aramoides Zelenkov [3]
Bumbaniralla walbeckornithoides Zelenkov [3]
Loncornis erectus Ameghino
Palaeogeranos tourmenti Louchart & Duhamel [2]
Papulavis annae Mourer-Chauviré et al.
Patagogrus olsoni Agnolín
Wanshuina lii Hou
Aptornithidae
Aptornis proasciarostratus Worthy, Tennyson & Scofield
Aramidae
Badistornis aramus Wetmore
Geranoididae
Eogeranoides campivagus Cracraft
Geranodornis aenigma Cracraft
Geranoides jepseni Wetmore
Palaeophasianus meleagroides Shufeldt
Paragrus prentici (Loomis)
Paragrus shufeldti Cracraft
Gruidae
Aramornis longurio Wetmore
Balearica excelsa Milne-Edwards
Balearica exigua Feduccia & Voorhies
Balearica rummeli Mlíkovský
Camusia quintanai Seguí
Eobalearica rinovi Gureev
Geranopsis hastingsiae Lydekker
Grus afghana Mourer-Chauviré et al.
Grus bogatshevi Serebrovakii
Grus cubensis Fischer & Stephan
Grus haydeni Marsh
Grus latipes Wetmore
Grus marshi Shufeldt
Grus melitensis Lydekker
Grus mongolica (Kurochkin)
Grus nannodes Wetmore & Martin
Grus pagei Campbell
Grus penteleci (Gaudry)
Grus primigenia Milne-Edwards
Grus sp. ‘Ryukuyu Islands, Japan’
Grus turfa Portis
Palaeogrus excelsa Milne-Edwards
Palaeogrus hordwelliensis Lydekker
Palaeogrus princeps (Portis)
Heliornithidae
Heliornithidae gen. & sp. ‘Pungo River Formation, USA’
Messelornithidae
Itardiornis hessae Mourer-Chauviré
Messelornis cristata Hesse,
Messelornis nearctica Hesse
Messelornis russelli Mourer-Chaviré
Pellornis mikkelseni Bertelli et al.
Walbeckornis creber Mayr
Parvigruidae
Alhuenia eduardotonnii Agnolín
Parvigrus pohli Mayr
Rupelrallus saxoniensis Fischer
Psophiidae
Archaeopsophia aoni Agnolín
Rallidae
Aletornis bellus Marsh
Aletornis nobilis Marsh
Aletornis pernix Marsh
Australlus disneyi (Boles)
Australlus gagensis Worthy & Boles
Baselrallus intermedius De Pietri & Mayr
Belgirallus minutus Mayr & Smith
Belgirallus oligocaenus Mayr & Smith
Creccoides osbornii Shufeldt
Crexica crexica Zelenkov, Panteleyev & De Pietri
Eocrex primus Wetmore
Fulica infelix Brodkorb
Fulica americana ssp. shufeldti Brodkorb
Fulica montanei Alarcón-Muñoz, Labarca & Soto-Acuña [1]
Fulica stekelesi Tchernov
Fulicaletornis venustus Marsh
Gallinula sp. ‘Csarnota, Ungarn’
Gallinula kansarum Brodkorb
Gallinula balcanica Boev
Ibidopsis hordwelliensis Lydekker
Latipons gardneri Harrison & Walker
Litorallus livezeyi Mather et al.
Miofulica dejardini Van Beneden
Miohypotaenidia tanaisensis Zelenkov, Panteleyev & De Pietri
Miorallus major Lambrecht
Palaeoaramides christyi (Milne-Edwards)
Palaeoaramides eximius Brodkorb
Palaeorallus troxelli Wetmore
Paraortygometra porzanoides Lambrecht
Pararallus dispar (Milne-Edwards)
Pararallus hassenkampi Martini
Pardirallus lacustris (Brodkorb)
Parvirallus gracilis Harrison & Walker
Pastushkinia zazhigini (Kurochkin)
Pleistorallus flemingi Worthy
“Porzana” botunensis Boev
“Porzana” estramosi Jánossy
“Porzana” kretzoii (Kessler)
Porzana payevskyi Zelenkov
“Porzana” piercei Olson & Wingate
“Porzana” risilla (Kurochkin)
“Porzana” sp. ‘QM F23253, Australien’
“Porzana” sp. ‘Văršec, Bulgarien’
“Porzana” veterior (Jánossy)
Priscaweka parvales Mather et al.
Quercyrallus arenarius Milne-Edwards
Quercyrallus dasypus Milne-Edwards
Quercyrallus ludianus Brodkorb
Rallicrex kolozsvarensis Lambrecht
Rallidae gen. & sp. ‘Chono-Harayah, Mongolei’
Rallidae gen. & sp. ‘Jamna Dolna, Polen’
Rallus auffenbergi Brodkorb
Rallus cyanocavi Steadman et al.
Rallus ibycus Olson & Wingate
Rallus natator Miller
Rallus philipsi Wetmore
Rallus polgardiensis (Jánossy)
Rallus prenticei Wetmore
Rallus recessus Olson & Wingate
Rallus richmondi Olson
Rallus sp. ‘Mátraszõlõs, Ungarn’
Rallus sp. ‘Ryukyu Islands, Japan’
Rallus sp. ‘Saw Rock Canyon, USA’
Rhenanorallus rhenanus Mayr
Youngornis gracilis Yeh
Youngornis qiluensis Yeh
Songziidae
Songzia acutunguis Wang, Mayr, Zhang & Zhou
Songzia heidangkouensis Hou
*********************
Quellen:
[1] Jhonatan Alarcón-Muñoz; Rafael Labarca; Sergio Soto-Acuña: Holocene rails (Gruiformes: Rallidae) of Laguna de Tagua Tagua Formation, central Chile, with the description of a new extinct giant coot. Journal of South American Earth Sciences 104. 2020
[2] Antoine Louchart; Anaïs Duhamel: A new fossil from the early Oligocene of Provence (France) increases the diversity of early Gruoidea and adds constraint on the origin of cranes (Gruidae) and limpkin (Aramidae). Journal of Ornithology 2021
[3] N. V. Zelenkov: New bird taxa (Aves: Galliformes, Gruiformes) from the early Eocene of Mogolia. Paleontological Journal 545(4). 2021
*********************
bearbeitet: 14.02.2024
Fossile Arten der Ordnung incertae sedis
Perplexicervicidae
‘Dynamopterus’ tuberculatus Peters [3]
Perplexicervix microcephalon Mayr [1]
Perplexicervix paucituberculata Mayr et al. [2]
Perplexicervix sp. ‘Quercy, Frankreich’ [3]
*********************
Quellen:
[1] Gerald Mayr: A new avian species with tubercle-bearing cervical vertebrae from the Middle Eocene of Messel (Germany). Records of the Australian Museum 62: 21-28. 2010
[2] Gerald Mayr; Vicen Carrió; Andrew C. Kitchener: On the “screamer-like” birds from the British London Clay: An archaic anseriform-galliform mosaic and a non-galloanserine “barb-necked” species of Perplexicervix. Palaeontologia Electronica 26(2): 1-23. 2023
[3] Gerald Mayr; Ursula B. Göhlich; Zbyněk Roček; Alfred Lemierre; Viola Winkler; Georgios L. Georgalis: Reinterpretation of tuberculate cervical vertebrae of Eocene birds as an exceptional anti‐predator adaptation against the mammalian craniocervical killing bite. journal of Anatomy 244: 402-410. 2024
*********************
bearbeitet: 24.04.2024
Fossil record of the Phaethontiformes
family incertae sedis
Clymenoptilon novaezealandicum Mayr et al.
Phaethonidae
Heliadornis ashbyi Olson
Phaethusavis pelagicus Bourdon et al.
Prophaethontidae
Lithoptila abdounensis Bourdon et al.
Prophaethon shrubsolei C. W. Andrews
Zhylgaia aestiflua Nessov
*********************
edited: 10.09.2023
Fossil order incertae sedis
Fossil order incertae sedis
Messelastur gratulator Peters
Tynskya brevitarsus Mayr & Kitchener [2]
Tynskya crassitarsus Mayr & Kitchener [2]
Tynskya eocaena Mayr
Tynskya waltonensis Mayr [1]
*********************
References:
[1] G. Mayr: A partial skeleton of a new species of Tynskya Mayr, 2000 (Aves, Messelasturidae) from the London Clay highlights the osteological distinctness of a poorly known early Eocene “owl/parrot mosaic”. PalZ advance online publication. doi: 10.1007/s12542-020-00541-8. 2021
[2] Gerald Mayr; Andrew C. Kitchener: The Vastanavidae and Messelasturidae (Aves) from the early Eocene London Clay of Walton-on-the-Naze (Essex, UK). Neues Jahrbuch für Geologie und Paläontologie 307(2): 113-139. 2023
*********************
edited: 02.08.2023
Fossil order incertae sedis
Halcyornithidae
Cyrilavis colburnorum Ksepka et al.
Cyrilavis olsoni Feduccia & Martin
Halcyornis toliapicus König
Pseudasturides macrocephalus (Mayr)
Pulchrapollia eximia Mayr & Kitchener
Pulchrapollia gracilis (Dyke & Cooper)
Pulchrapollia tenuipes Mayr & Kitchener
Serudaptus pohli Mayr
*********************
edited: 02.08.2023
Fossile Psittaciformes
Cacatuidae
Cacatua sp. ‘Riversleigh, Australien’
Psittacidae
Agapornis atlanticus Mourer-Chauviré
Agapornis attenboroughi Manegold
Agapornis sp. ‘Kromdraai B, Südafrika’
Agapornis sp. ‘Plovers Lake, Südafrika’
Aratinga roosevelti Spillman
Aratinga vorohuensis (Tonni & Noriega)
Archaeopsittacus verreauxi Milne-Edwards
Bavaripsitta ballmanni Mayr & Göhlich
Conuropsis fratercula Wetmore
Khwenena leopoldinae Manegold
Melopsittacus undulatus (ssp. ‘Pliozän’ ?)
Mogontiacopsitta miocaena Mayr
Psittacidae gen. & sp. ‘Baikalsee, Russland’
Xenopsitta feifari Mlíkovsky
Quercypsittidae
Quercypsitta ivani Mourer-Chauviré
Quercypsitta sp. ‘Walton-on-the-Naze, Großbritannien’
Quercypsitta sudrei Mourer-Chauviré
Strigopidae
Heracles inexpectatus Worthy, Hand, Archer, Scofield & De Pietri
Nelepsittacus daphneleeae Worthy et al.
Nelepsittacus donmertoni Worthy et al.
Nelepsittacus minimus Worthy et al.
Nelepsittacus (?) sp. ‘Croc Site Layer, Neuseeland’
*********************
bearbeitet: 19.04.2024
Mo’ora-‘ura and toroa – a red duck and a surf duck from Tahiti?
In ancient times, Tahitians believed that several of their Gods and other celestial beings would show themselves to the human eye in form of an animal, a so-called ata and Teuira Henry [see also here] lists some of them, among them also three kinds of ducks.:
“Red-feathered duck (mo‘ora-‘ura), ata of ‘Orovehi‘ura (‘Oro in his manifestation of Red-feather-covered).
Wild duck (mo‘ora-ōviri), ata of “sylvan elves.”
Surf duck (toroa), ata of Hau, god of peace.” [1][3]
The ‘Wild duck’, mentioned here, is the Pacific Black Duck (Anas superciliosa J. F. Gmelin) which is the only duck species known from French Polynesia at all, so what are the other two ‘ducks’?
“The most showy headdress worn officially by the king and princes and high chiefs was the taumi, a superb helmet made of clusters of crimson feathers of the moora ‘ura (red-feathered duck), set upon a light framework and covering the head like a bird, with a glossy terminal behind of outspreading red, black, and white feathers tastily mixed together.” [1][3]
A taumi, however, actually is a gorget decorated with feathers, a feathered helmet was called fau.:
“Henry gives a description of a headdress which has some characteristics of a fau, but which seems to be a mixture of remembered types, further confused by the use of the name taumi, which we know was a gorget:” [4]
The fau was also decorated with several of the elongated tail feathers of tropicbirds (as you can see in the depiction, in which they are from the White-tailed Tropicbird (Phaethon lepturus ssp. dorotheae Daudin)).
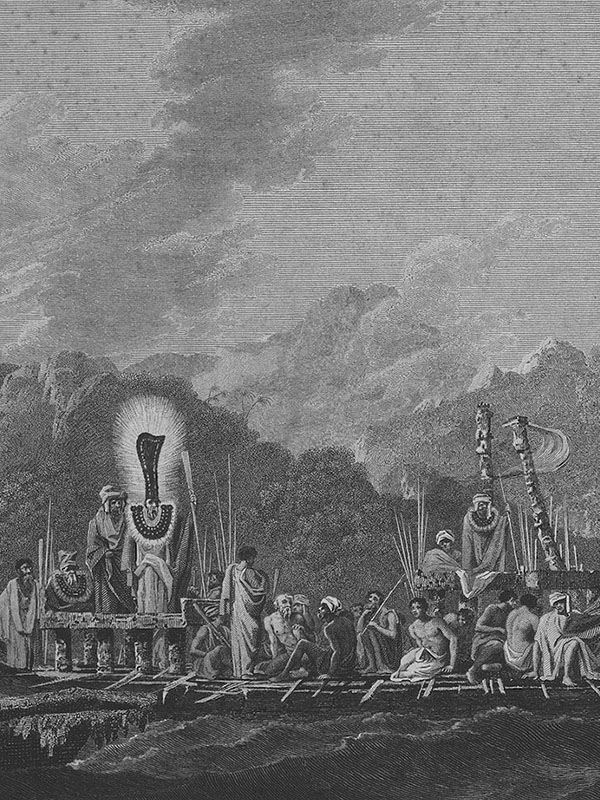
Depiction from: ‘William Hodges: The fleet of Otaheite assembled at Oparee, 1777’
(public domain)
*********************
A little update here:
A “surf duck” apparently is nothing but an albatross (Diomedea spp.), a bird that is well known to the Polynesians, even in the tropical parts of the region. [2]
*********************
References:
[1] Teuira Henry: Ancient Tahiti. Bishop Museum Bulletins 48: 1-651. 1928
[2] Kenneth P. Emory: Tuamotuan bird names. The Journal of the Polynesian Society 56(2): 188-196. 1947
[3] Douglas L. Oliver: Ancient Tahitian Society. The University Press of Hawai’i, Honolulu 1974
[4] Karen Stevenson; Steven Hooper: Tahitian fau – unveiling an enigma. Journal of the Polynesian Society 116(2): 181-212. 2007
*********************
edited: 31.07.2023
A (not really) new species of sunbird – Wakatobi Subird (Cinnyris infrenatus Hartert)
The Wakatobi Sunbird was actually described in 1903; it was subsequently reclassified as a subspecies of the Sahul Sunbird (Cinnyris clementiae Lesson) or the Olive-backed Sunbird (Cinnyris jugularis L.); it was only recently recognized as being a distinct species (again). [1][2]
*********************

https://www.inaturalist.org/people/abuhamas
http://creativecommons.org/licenses/by-sa/4.0/
*********************
References:
[1] Darren P. O’Connell; David J. Kelly; Seán B. A. Kelly; Kangkuso Analuddin; Adi Karya; Nicola M. Marples; Frank E. Rheindt; Thomas E. Martin: An assessment of the avifauna of the Wakatobi Islands, South-east Sulawesi, Indonesia: species recorded and taxonomic considerations. Raffles Bulletin of Zoology 68: 574-587. 2020
[2] Fionn Ó Marcaigh; David J Kelly; Darren P O’Connell; Kangkuso Analuddin; Adi Karya; Jennifer McCloughan; Ellen Tolan; Naomi Lawless; Nicola M Marples: Small islands and large biogeographic barriers have driven contrasting speciation patterns in Indo-Pacific sunbirds (Aves: Nectariniidae). Zoological Journal of the Linnean Society 198(1): 72-92. 2023
*********************
edited: 21.07.2023
A new species of oriole – Ventriloquial Oriole (Oriolus consobrinus Ramsay)
The Dark-throated Oriole (Oriolus xanthonotus Horsfield) was thought to include four subspecies inhabiting parts of Southeast Asia including some islands of Indonesia as well as the island of Palawan north of Borneo; however, according to a recent study, it is now known that some of the populations are not closely related to each other at all.
Birds from northern Borneo and Palawan are more closely related to the Philippine oriole species and therefore should be treated as a distinct taxon for which the name Ventriloquial Oriole (Oriolus consobrinus Ramsay) has been suggested.
The new species is superficially identical to the Dark-throated Oriole but is somewhat smaller and can be distinguished by its song. [1]
*********************

Photo: bejamynweil
https://www.inaturalist.org/people/benjamynweil
https://creativecommons.org/licenses/by-nc/4.0/

Photo: Forest Botial-Jarvis
https://www.inaturalist.org/people/tiluchi
https://creativecommons.org/licenses/by-nc/4.0/
*********************
References:
[1] Frank E. Rheindt; Meng Yue Wu; Nyanasengeran Movin; Knud A. Jønsson: Cryptic species-level diversity in Dark-throated Oriole Oriolus xanthonotus. Bulletin of the British Ornithologists’ Club 142(2): 254-267. 2022
*********************
edited:
20.07.2023
A new subspecies of the Ruby-throated Bulbul
The Ruby-throated Bulbul (Rubigula dispar (Horsfield)) is known to inhabit the islands of Bali, Java and Sumatra in Indonesia and was until now considered monotypic, that means the populations were not seen as being distinct in any way.
According to a new study, however, it is now known that the birds from Sumatra differ from the others by their bright red instead of yellow eyes, and to some degree by subtle differences of the color of their plumage as well as by having a slightly longer beak.
The Sumatran birds were accordingly classified as a new subspecies and named as Rubigula dispar ssp. matamerah Berryman & Collar.
*********************

Photo: Bruno Durand
https://www.inaturalist.org/people/brunodurand
https://creativecommons.org/licenses/by-nc/4.0/

Photo: Mitch Rose
https://www.inaturalist.org/people/mroseup
https://creativecommons.org/licenses/by-nc/4.0/
*********************
References:
[1] Alex Berryman; Nigel J. Collar: Subspeciation in the Ruby-throated Bulbul Rubigula dispar. Bulletin of the British Ornithologists’ Club 143(2): 237-243. 2023
*********************
edited: 19.07.2023
From one to seven – Slaty-backed Nightingale-Thrush
The Slaty-backed Nightingale-Thrush (Catharus fuscater (Lafresnaye)) is a rather small, slaty grey colored thrush that looks as if it’s constantly judging you; it inhabits dense forests in the northwestern parts of South America.
The species, despite being quite common and widespread, is not easy to observe and thus not well-known.
According to a new study, the Slaty-backed Nightingale-Thrush is in fact is a species complex containing at least seven species as well as four subspecies, some of them newly described. [1]
***
Unfortunately, I have not yet access to this study, so I do not know more about it.
*********************

https://creativecommons.org/licenses/by-sa/4.0/
*********************
References:
[1] Matthew R. Halley; Therese A Catanach; John Klicka; Jason D Weckstein: Integrative taxonomy reveals hidden diversity in the Catharus fuscater (Passeriformes: Turdidae) complex in Central and South America. Zoological Journal of the Linnean Society. advance online publication. doi: 10.1093/zoolinnean/zlad031. 2023
*********************
edited: 08.07.2023
Did parakeets once inhabit the Tuamotu Islands?
Parakeets of the genus Cyanoramphus are known to once have existed in French Polynesia, namely on the Austral- and the Society Islands. Is it possible that one or more species of that genus once also have existed on the Tuamotu Archipelago?
Well, actually, it absolutely is, but there is yet no actual indication of it; or maybe there is one …:
In a count of Tuamotuan bird names from the 1940s the name petea is given as coming “from an informant in the western Tuamotus, who said it was like the petea of Tahiti.” [1] and that is probably the name of a parakeet. Maybe this informant just gave that name and told the author that it is given to a long-tailed bird that is also found on Tahiti, who knows?
Well, a short check informs us that petea or rather pete’a is indeed a Tahitian bird name but actually for the White-tailed Tropicbird (Phaethon lepturus ssp. dorotheae Mathews) which in fact is not a parakeet, unfortunately.
***
So, while the former occurrence of parakeets on at least some of the atolls and islands in the giant Tuamotuan Archipelago seems absolutely possible, we have yet no real proof of that … and probably never will.
But we can let our imagination run wild, why not?!
*********************
References:
[1] Kenneth P. Emory: Tuamotuan bird names. The Journal of the Polynesian Society 56(2): 188-196. 1947
*********************
edited: 07.07.2023
Acrocephalus sp. BMNH 1846.7.29.6
The British Museum of Natural History is apparently housing a reed-warbler specimen that is sister to the Leeward Islands Reed-Warbler (Acrocephalus musae (J. R. Forster)) , a now extinct species that occurred with two subspecies on the islands of Huahine and Ra’iatea in the leeward group of the Society Islands.
However, this single specimen appears not to be identical to either of the two known subspecies and was proposed to be classified as “A. musae subsp. (Leeward Islands), with uncertainty regarding the island and subspecies.” [1]; it might indeed represent another, third subspecies that had been collected from another island within the leeward group.
*********************
References:
[1] Alice Cibois; Jean-Claude Thibault; Eric Pasquet: Molecular and morphological analysis of Pacific reed warbler specimens of dubious origin, including Acrocephalus luscinius astrolabii. Bulletin of the British Ornithologist’s Club 131(1): 32-40. 2011
*********************
edited: 05.07.2023
The hypothetical birds of Polynesia
… just a list (in alphabetical order) of the hypothetical birds (marked in green letters) that once may have existed within the Polynesian triangle, in a rather conservative approach!
***
Acrocephalidae
Acrocephalus kerearako ssp. ”Atiu’
Acrocephalus kerearako ssp. ‘Ma’uke’
Acrocephalus musae ssp. ‘Bora Bora’
Acrocephalus musae ssp. ‘Maupiti’
Acrocephalus sp. ‘Austral Islands’
Acrocephalus sp. ‘Palmyra Atoll’
Alcedinidae
Todiramphus sp. ‘Makatea’
Todiramphus sp. ‘Me’eti’a’
Todiramphus sp. ‘Raivavae’
Todiramphus sp. ‘Rimatara’
Todiramphus sp. ‘Rurutu’
Todiramphus sp. ‘Tubuai’
Todiramphus tutus ssp. ‘Miti’aro’
Todiramphus ‘veneratus ssp.’ ‘Huahine’
Todiramphus ‘veneratus ssp.’ ‘Ra’iatea’
Anatidae
Anas sp. ‘Kiritimati’
Anatidae gen. & sp. ‘Ni’ihau Moa nalo’
Apodidae
Aerodramus sp. ‘Austral Islands’
Aerodramus sp. ‘Ma’uke’
Aerodramus sp. ‘Miti’aro’
Aerodramus sp. ‘Rarotonga’
Ardeidae
Butorides striata ssp. ‘Austral Islands’
Butorides striata ssp. ‘Cook Islands’
Butorides striata ssp. ‘Samoa’
Butorides striata ssp. ‘Tonga’
Nycticorax sp. ‘Samoa’ – may have survived on the Manu’a Islands (probably Ta’u) until the 1930s [3]
Columbidae
Ducula sp. ‘Pitcairn Island’
Macropygia sp. ‘Cook Islands’
Macropygia sp. ‘Fiji’
Macropygia sp. ‘Samoa’
Macropygia sp. ‘Tonga’
Pampusana sp. ‘Pitcairn Island’ [4]
Pampusana sp. ‘Raivavae’ [4]
Pampusana sp. ‘Rapa’ [4]
Pampusana sp. ‘Rimatara’ [4]
Ptilinopus sp. ‘Pitcairn Island’
Estrildidae
Erythrura sp. ‘Rotuma’
Erythrura sp. ‘Tonga’
Erythrura sp. ‘Wallis & Futuna’
Fringillidae
Chloridops sp. ‘Ni’ihau’
Hemignathus sp. ‘Ni’ihau’
Hemignathus sp. ‘Ni’ihau Akialoa’
Hemignathus sp. ‘Ni’ihau Nukupu’u’
Loxioides sp. ‘Ni’ihau’
Loxops sp. ‘Ni’ihau’
Magumma sp. ‘Ni’ihau’
Oreomystis sp. ‘Ni’ihau’
Paroreomyza montana ssp. ‘Kaho’olawe’
Rhodacanthis sp.’Ni’ihau’
Telespiza sp. ‘Ni’ihau’
Xestospiza sp. ‘Ni’ihau’
Hemiprocnidae
Hemiprocne (mystacina) ssp. ‘Fiji’
Hirundinidae
Hirundo javanica ssp. ‘Samoa’
Hirundo sp. ‘Austral Islands’
Hirundo sp. ‘Cook Islands’
Hirundo sp. ‘Mangareva’
Lamproliidae
Lamprolia sp. ‘Kadavu’
Lamprolia sp. ‘Viti Levu’
Locustellidae
Megalurulus rufa ssp. ‘Kadavu’
Megalurulus rufa ssp. Taveuni
Megapodiidae
Megapodius sp. ‘Rotuma’
Megapodius sp. ‘Samoa’
Megapodius sp. ‘Wallis & Futuna’
Meliphagidae
Gymnomyza sp. ‘Kadavu’
Myzomela sp. ‘Futuna’
Myzomala sp. ”Uvea’
Mohoidae (endemic family)
Moho sp. ‘Ni’ihau’
Monarchidae
Monarcha sp. ‘Bora Bora’
Monarcha sp. ‘Huahine’
Monarcha sp. ‘Makatea’
Monarcha sp. ‘Mangaia’
Monarcha sp. ‘Maui Nui’
Monarcha sp. ‘Mo’orea’
Monarcha sp. ‘Ni’ihau’
Monarcha sp. ‘Ra’iatea/Taha’a’
Myiagra sp. ‘Rotuma’
Myiagra sp. ‘Tongatapu’
Motacillidae
Anthus novaeseelandiae ssp. ‘Macquarie Island’
Pachycephalidae
Pachycephala jacquinoti ssp. ‘Ha’apai Islands’
Pachycephala jacquinoti ssp. ‘Tongatapu’
Pandionidae
Pandion cristatus ssp. ‘Fiji’
Petroicidae
Petroica pusilla ssp. ”Eua’
Petroica pusilla ssp. ‘Tongatapu’
Petroica pusilla ssp. ‘Vava’u Islands’
Petroica pusilla ssp. ‘Wallis & Futuna’
Psittacidae
Cyanoramphus sp. ‘Bora Bora’
Cyanoramphus sp. ‘Cook Islands’
Cyanoramphus sp. ‘Huahine’
Cyanoramphus sp. ‘Raivavae’
Cyanoramphus sp. ‘Rimatara’
Cyanoramphus sp. ‘Rurutu’
Cyanoramphus sp. ‘Tubuai’
Cyanoramphus zealandicus ssp. ‘Mo’orea’
Eclectus infectus ssp. ‘Fiji’
Vini kuhlii ssp. ‘Society Islands’ – may have survived on Bora Bora until the 1830s [1]
Vini sp. ‘Rapa’
Rallidae
Fulica sp. ‘Fiji’
Gallinula sp. ‘Tonga’
Gallirallus sp. ‘Aitutaki’
Gallirallus sp. ‘Alofi’
Gallirallus sp. ”Atiu’
Gallirallus sp. ‘Bora Bora’
Gallirallus sp. ‘Fatu Hiva’
Gallirallus sp. ‘Futuna’
Gallirallus sp. ‘Mai’ao’
Gallirallus sp. ‘Makatea’
Gallirallus sp. ‘Ma’uke’
Gallirallus sp. ‘Miti’aro’
Gallirallus sp. ‘Mo’orea’
Gallirallus sp. ‘Niau’
Gallirallus sp. ‘Ra’iatea’
Gallirallus sp. ‘Raivavae’
Gallirallus sp. ‘Rarotonga’
Gallirallus sp. ‘Rimatara’
Gallirallus sp. ‘Rotuma’
Gallirallus sp. ‘Savai’i’
Gallirallus sp. ‘Taveuni’
Gallirallus sp. ‘Ua Pou’
Gallirallus sp. ”Uvea’
Gallirallus sp. ”Upolu’
Porphyrio sp. ‘Bora Bora’
Porphyrio sp. ‘Fatu Hiva’
Porphyrio sp. ‘Mo’orea’
Porphyrio sp. ‘Nuku Hiva’
Porphyrio sp. ‘Rarotonga’
Porphyrio sp. ‘Tahiti’
Porphyrio sp. ‘Ua Huka’
Porphyrio sp. ‘Ua Pou’
Zapornia sp. ‘Aitutaki’
Zapornia sp. ‘Alofi’
Zapornia sp. ‘Anaa’
Zapornia sp. ”Atiu’
Zapornia sp. ‘Bora Bora’
Zapornia sp. ‘Eiao’
Zapornia sp. ‘Fatu Hiva’
Zapornia sp. ‘Futuna’
Zapornia sp. ‘Hiva Oa’
Zapornia sp. ‘Huahine’
Zapornia sp. ‘Kaho’olawe’
Zapornia sp. ‘Lana’i’
Zapornia sp. ‘Mai’ao’
Zapornia sp. ‘Makatea’
Zapornia sp. ‘Mangareva’
Zapornia sp. ‘Ma’uke’
Zapornia sp. ‘Maupiti’
Zapornia sp. ‘Me’eti’a’
Zapornia sp. ‘Miti’aro’
Zapornia sp. ‘Mohotani’
Zapornia sp. ‘Mo’orea’
Zapornia sp. ‘Niau’
Zapornia sp. ‘Ni’ihau’
Zapornia sp. ‘Pitcairn Island’
Zapornia sp. ‘Ra’iatea’
Zapornia sp. ‘Raivavaea’
Zapornia sp. ‘Rapa’
Zapornia sp. ‘Rarotonga’
Zapornia sp. ‘Rimatara’
Zapornia sp. ‘Rotuma’
Zapornia sp. ‘Rurutu’
Zapornia sp. ‘Savai’i’
Zapornia sp. ‘Taha’a’
Zapornia sp. ‘Tahuata’
Zapornia sp. ‘Tetiaroa’
Zapornia sp. ‘Tubuai’
Zapornia sp. ‘Tupai’
Zapornia sp. ‘Ua Pou’
Zapornia sp. ”Upolu’
Zapornia sp. ”Uvea
Rhipiduridae
Rhipidura sp. ”Eua’
Rhipidura sp. ‘Tongatapu’
Scolopacidae
Coenocorypha sp. ‘Kermadec Islands’
Coenocorypha sp. ‘Macquarie Island’
Strigidae
Grallistrix sp. ‘Lana’i’
Grallistrix sp. ‘Ni’ihau’
Sturnidae
Aplonis sp. ‘Austral Islands‘
Aplonis sp. ‘Bora Bora’
Aplonis sp. ‘Mangaia’ – may have survived into the late 19th/early 20th century [2]
Aplonis sp. ‘Mai’ao’
Aplonis sp. ‘Maupiti’
Aplonis sp. ‘Mo’orea’
Aplonis sp. ‘Tahiti’
Turdidae
Myadestes lanaiensis ssp. ‘Kaho’olawe’
Myadestes sp. ‘Ni’ihau’
Turdus sp. ‘Wallis & Futuna’
Zosteropidae
Zosteropidae gen. & sp. ‘Samoa’
***
Let’s see which of ‘my’ hypothetical birds will be discovered in the future. 😛
*********************
References:
[1] M. L. I. Duperrey: Voyage autour du monde: Exécuté par Ordre du Roi, Sur la Corvette de Sa Majesté, La Coquille, pendant les années 1822, 1823, 1824, et 1825, par M. L. I. Duperrey; Zoologie, par Mm. Lesson et Garnot. Paris: Arthus Bertrand 1828
[2] F. W. Christian: List of Mangaia birds. The Journal of the Polynesian Society 29(114): 87. 1920
[3] J. S. Armstrong: Hand-list to the Birds of Samoa. John Bale, Sons and Danielsson, London 1932
[4] M. Bruce; N. Bahr; N. David: Pampusanna vs. Pampusana: a nomenclatural conundrum resolved, along with associated errors and oversights. Bulletin of the British Ornithologists’ Club. 136: 86–100. 2016
*********************
edited: 29.06.2023
Neuntöter

*********************
bearbeitet: 29.06.2023
Buchfink

*********************
bearbeitet: 29.06.2023
Fossile Sphenisciformes
Spheniscidae
Anthropodyptes gilli Simpson
Anthropornis grandis Wiman
Anthropornis nordenskjoeldi Wiman
Aprosdokitos mikrotero Acosta-Hospitaleche et al.
Aptenodytes ridgeni Simpson
Apterodytes ictus Ameghino
Archaeospheniscus lopdelli Marples
Archaeospheniscus lowei Marples
Archaeospheniscus wimani Marples
Arthrodytes andrewsi Ameghino
Crossvallia unienwillia Tambussi et al.
Crossvallia (?) waiparensis Mayr, De Pietri, Love, Mannering & Scofield
Dege hendeyi Simpson
Delphinornis arctowskii Myrcha et al.
Delphinornis larseni Wiman
Duntroonornis parvus Marples
Eudyptes atatu Thomas, Tennyson, Scofield, Heath, Pett & Ksepka [1]
Eudyptula wilsonae Thomas et al.
Icadyptes salasi Clarke et al.
Inguza predemersus Simpson
Kaiika maxwelli Fordyce & Thomas
Kairuku grebneffi Ksepka, Fordyce, Ando & Jones
Kairuku waitaki Ksepka, Fordyce, Ando & Jones
Kairuku waewaeroa Giovanardi, Ksepka & Thomas [2]
Korora oliveri Marples
Kumimanu biceae Mayr et al.
Kumimanu fordycei Ksepka et al.
Kupoupou stilwelli Blokland, Reid, Worthy, Tennyson, Clarke & Scofield
Madrynornis mirandus Acosta-Hospitaleche et al.
Marambiornis exilis Myrcha et al.
Marambiornopsis sobrali Jadwiszczak et al. [3]
Marplesornis novaezealandiae Marples
Mesetaornis polaris Myrcha et al.
Muriwaimanu tuatahi Slack et al.
Nucleornis insolitus Simpson
Orthopteryx gigas Wiman
Pachydyptes ponderosus Oliver
Pachydyptes simpsoni Jenkins
Palaeeudyptes antarcticus Huxley
Palaeeudyptes gunnari Wiman
Palaeeudyptes klekowskii Myrcha et al.
Palaeeudyptes marplesi Brodkorb
Paraptenodytes antarcticus Moreno & Mercerat
Paraptenodytes brodkorbi Simpson
Paraptenodytes robustus Ameghino
Perudyptes devriesi Clarke et al.
Petradyptes stonehousei Ksepka et al.
Platydyptes amiesi Marples
Platydyptes marplesi Simpson
Platydyptes novaezealandiae Oliver
Pseudaptenodytes macraei Simpson
Pseudaptenodytes minor Simpson
Pygoscelis calderensis Acosta-Hospitaleche, Chávez & Fritis
Pygoscelis grandis Walsh & Suarez
Pygoscelis tyreei Simpson
Sequiwaimanu rosieae Mayr et al.
Spheniscidae gen. & sp. ‘Burnside Formation, Neuseeland’
Spheniscus chilensis Emslie & Correa
Spheniscus megaramphus Stucchi et al.
Spheniscus muizoni Gohlich
Spheniscus urbinai Stucchi
Tonniornis mesetaensis Tambussi et al.
Tonniornis minimum Tambussi et al.
Waimanu manneringi Slack, Jones, Ando, Harrison, Fordyce, Arnason & Penny
Wimanornis seymourensis Simpson
*********************
Referenzen:
[1] Daniel B. Thomas; Alan J. D. Tennyson; R. Paul Scofield; Tracy A. Heath; Walker Pett; Daniel T. Ksepka: Ancient crested penguin constrains timing of recruitment into seabird hotspot. Proceedings of the Royal Society B. 287(1932), 2020, s. 20201497, DOI: 10.1098/rspb.2020.1497
[2] Simone Giovanardi; Daniel T. Ksepka; Daniel B. Thomas: A giant Oligocene fossil penguin from the North Island of New Zealand. Journal of Vertebrate Paleontology: e1953047. doi:10.1080/02724634.2021.1953047. 2021
[3] P. Jadwiszczak; M. Reguero; T. Mörs: A new small-sized penguin from the late Eocene of Seymour Island with additional material of Mesetaornis polaris. GFF advance online publication. doi: 10.1080/11035897.2021.1900385. 2021
*********************
edited: 22.06.2023
Fossile Suliformes
Anhingidae
Anhinga beckeri Emslie
Anhinga fraileyi Campbell
Anhinga hadarensis Brodkorb & Mourer-Chauviré
Anhinga pannonica Lambrecht
Anhinga subvolans (Brodkorb)
Anhinga walterbolesi Worthy
Macranhinga paranensis Noriega
Meganhinga chilensis Alvarenga
Fregatidae
Limnofregata azygosternon Olson
Limnofregata hasegawai Olson & Matsuoka
Limnofregata hutchisoni Stidham
Phalacrocoracidae
Borvocarbo guilloti Mourer-Chauviré
Borvocarbo stoeffelensis Mayr
Borvocarbo tardatus Gohlich & Mourer-Cauviré
Limicorallus saiensis Kurochkin
Oligocorax littoralis Lambrecht
Oligocorax miocaenus Milne-Edwards
Paracorax destefanii Regalia
Phalacrocorax anatolicus Paicheler et al.
Phalacrocorax chapalensis Alvarez
Phalacrocorax femoralis Miller
Phalacrocorax filyawi Emslie
Phalacrocorax goletensis Howard
Phalacrocorax idahensis Marsh
Phalacrocorax intermedius Milne-Edwards
Phalacrocorax kennelli Howard
Phalacrocorax leptopus Brodkorb
Phalacrocorax longipes Tugarinov
Phalacrocorax macer Brodkorb
Phalacrocorax macropus Cope
Phalacrocorax marinavis Shufeldt
Phalacrocorax mediterraneus Shufeldt
Phalacrocorax mongoliensis Kurochkin
Phalacrocorax novaezealandiae Forbes
Phalacrocorax praecarbo von Ammon
Phalacrocorax reliquus Kurochkin
Phalacrocorax risgoviensis Fraas
Phalacrocorax rogersi Howard
Phalacrocorax wetmorei Brodkorb
Piscator tenuirostris Harrison & Walker
Praecarbo strigoniensis Kessler & Horváth
Stictocarbo kumeyaay Chandler
Plotopteridae
Copepteryx hexeris Olson & Hasegawa
Copepteryx titan Olson & Hasegawa
Empeirodytes okazakii Ohashi & Hasegawa [1]
Hokkaidornis abashiriensis Sakurai et al.
Klallamornis abyssa Mayr & Goedert
Klallamornis clarki Mayr & Goedert
Phocavis maritimus Goedert
Plotopterum joaquinensis Howard
Stemec suntokum Kaiser et al.
Stenornis kanmonensis Ohashi & Hasegawa [1]
Tonsala buchanani Dyke et al.
Tonsala hildegardae Olson
Protoplotidae
Protoplotus beauforti Lambrecht
Sulidae
Bimbisula melanodactylos Benson & Erickson
Empheresula arvernensis Milne-Edwards
Eostega lebedinskyi Lambrecht
Masillastega rectirostris Mayr
Microsula pygmaea Milne-Edwards
Miosula media Miller
Morus atlanticus Shufeldt
Morus avitus Wetmore
Morus humeralis Miller & Bowman
Morus lompocanus Miller
Morus loxostylus Cope
Morus magnus Howard
Morus peninsularis Brodkorb
Morus peruvianus Stucchi
Morus recentior Howard
Morus reyana Howard
Morus vagabundus Wetmore
Paleosula stocktoni Miller
Prophalacrocorax ronzoni Gervais
Ramphastosula aguierrei Stucchi et al.
Ramphastosula ramirezi Stucchi & Urbina
Sarmatosula dobrogensis Grigorescu & Kessler
Sula arvernensis Milne-Edwards
Sula brandi Stucchi et al.
Sula clarki Chandler
Sula figueroae Stucchi et al.
Sula guano Brodkorb
Sula magna Stucchi
Sula phosphata Brodkorb
Sula pohli Howard
Sula sulita Stucchi
Sula universitatis Brodkorb
Sula willetti Miller
*********************
Quelle:
[1] Tomoyuki Ohashi; Yoshikazu Hasegawa: New species of Plotopteridae (Aves) from the Oligicene Ashiya Group of northern Kyushu, Japan. Paleontological Research 24(4): 285-297. 2020
*********************
bearbeitet: 17.06.2023
Mäusebussard

*********************
bearbeitet: 29.06.2023
Tafelenten


*********************
bearbeitet: 29.06.2023
Nilgans

*********************
bearbeitet: 29.06.2023
Höckerschwäne

*********************
bearbeitet: 29.06.2023
Fossile Arten der Pelecaniformes
Ardeidae
Ardea aurelianensis Milne-Edwards
Ardea brunhuberi von Ammon
Ardea effosa von Meyer
Ardea formosa Milne-Edwards
Ardea latipes von Meyer
Ardea lignitum Gibel
Ardea paloccidentalis Shufeldt
Ardea perplexa Milne-Edwards
Ardea piveteaui Brunet
Ardea polkensis Brodkorb
Ardeagradis arborea Autor ?
Egretta subfluvia Becker
Gnotornis aramiellus Wetmore
Matuku otagoense Scofield et al.
Nycticorax fidens Brodkorb
Nycticorax sp. ‚Fayyum, Ägypten‘
Palaeophoyx columbiana McCoy
Pikaihao bartlei Worthy et al.
„Proardea“ deschutteri Mayr, De Pietri, Scofield & Smith
Proardeola walkeri Harrison
Zeltornis ginsburgi Balouet [1]
Pelecanidae
Eopelecanus aegyptiacus El Adli et al. [2]
Pelecanus cadimurka Rich & Van Tets
Pelecanus cautleyi Davies
Pelecanus fraasi Lyddeker
Pelecanus gracilis Milne-Edwards
Pelecanus grandiceps Des Vis
Pelecanus halieus Wetmore
Pelecanus intermedius Fraas
Pelecanus odessanus Widhalm
Pelecanus paranensis Noriega et al.
Pelecanus proavus De Vis
Pelecanus schreiberi Olson
Pelecanus sivalensis Davies
Pelecanus tirarensis Miller
Threskiornithidae
Actiornis anglicus Lydekker
Ajaja chione Emslie
Eudocimus leiseyi Emslie
Eudocimus peruvianus Campbell
Gerandibis pagana (Milne-Edwards)
Geronticus cf. calvus (Boddaert) ‚Gauteng, Südafrika‘
Ibidopodia palustris Milne-Edwards
Milnea gracilis Lydekker
Minggangia changgouensis Hou
Protibis cnemialis Ameghino
Rhynchaeites messelensis Wittich
Rhynchaeites litoralis Mayr & Kitchener
Rhynchaeites sp. ‚Fur Formation, Dänemark‘
Rhynchaeites tanta (Waterhouse et al.)
Sanshuiornis zhangi Wang, Mayr, Zhang & Zhou
Vadaravis brownae Smith, Grande & Clarke
*********************
Quellen:
[1] Jiří Mlíkovský: Early Miocene birds of Djebel Zelten, Libya. Časopis Národního muzea, Řada přírodovědná 172(1-4): 14-120. 2003
[2] Joseph J. El Adli; Jeffrey A. Wilson Mantilla; Mohammed Sameh M. Antar; Philip D. Gingerich: The earliest recorded fossil pelican, recovered from the late Eocene of Wadi Al-Hitan, Egypt. Journal of Vertebrate Paleontology 2021. DOI: 10.1080/02724634.2021.1903910
*********************
bearbeitet: 24.04.2024
Fossile Trogoniformes
Familie incertae sedis (?)
Foshanornis songi Zhao, Mayr, Wang & Wang
Trogonidae
Eotrogon stenorhynchus Mayr, De Pietri & Kitchener
Masillatrogon pumilio Mayr
Paratrogon gallicus Milne-Edwards
Primotrogon (?) sp. ‘Steendorp, Belgien’
Primotrogon wintersteini Mayr
Septentrogon madseni Kristoffersen
Trogonidae gen. & sp. ‘Matt, Schweiz’
*********************
bearbeitet: 10.05.2023
Hausspatzen


*********************
bearbeitet: 29.06.2023
Wiedehopf

*********************
bearbeitet: 29.06.2023
Kolkraben

*********************
bearbeitet: 29.06.2023
Scotocercidae – Wüstendickichtsänger
Diese kleine Familie von unauffälligen wüstenbewohnenden Vögelchen umfasst, je nach Ansicht, eine oder zwei Arten mit insgesamt acht Unterarten, die im Norden Afrikas, in Teilen Arabiens sowie im südlichen Westasien verbreitet sind.
***
Die Darstellung zeigt die einzigen beiden Arten der Familie, den Wüstendickichtsänger (Scotocerca inquieta (Cretzschmar)) (unten) und den Saharadickichtsänger (Scotocerca saharae Loche) (oben).
*********************

(public domain)
*********************
Quellen:
[1] Per Alström; Jon Fjeldså; Silke Fregin; Urban Olsson: Gross morphology betrays phylogeny: the Scrub Warbler Scotocerca inquieta is not a cisticolid. The Ibis 153(1): 87-97. 2011
[2] Silke Fregin; Martin Haase; Urban Olsson; Per Alström: New insights into family relationships within the avian superfamily Sylvioidea (Passeriformes) based on seven molecular markers. BMC Evolutionary Biology 12(1): 1-12. 2012
*********************
bearbeitet: 13.04.2023
Fossile Passeriformes
Familie incertae sedis
Certhiops rummeli Manegold
Corvitalusoides grandiculus Boles
Crosnoornis nargizia Bochenski, Tomek, Bujoczek & Salwa [11]
Jamna szybiaki Bocheński, Tomek, Bujoczek & Wertz
Kischinskinia scandens Volkova & Zelenkov
Minutornis primoscenoides May & Kitchener
Resoviaornis jamrozi Bocheński, Tomek, Wertz & Świdnicka
Sylvosimadaravis janossyi (Kessler et Hír)
Winnicavis gorskii Bocheński, Tomek, Wertz, Happ, Bujoczek & Świdnicka
Acanthisittidae
Kuiornis indicator Worthy et al.
Acanthizidae
Acanthizidae gen. & sp. ‘Riversleigh, Australien’
Acrocephalidae
Acrocephalus major Kessler
Acrocephalus minor Kessler
Acrocephalus kordosi Kessler
Acrocephalus kretzoii Kessler
Acrocephalus sp. ‘Riversleigh, Australien’
Hippolais veterior Kessler
Aegithalidae
Aegithalos congruis Kessler
Aegithalos gaspariki Kessler
Alaudidae
Alauda trivadari Kessler
Alauda xerarvensis Boev [2]
Ammomanes prealpestris (Boev)
Calandrella gali Kessler
Eremarida xerophila Boev [2]
Eremophila alpestris ssp. [6]
Eremophila orkhonensis (Zelenkov & Kurochkin)
Galerida bulgarica Boev [2]
Galerida cserhatensis Kessler & Hír
Galerida pannonica Kessler
Lullula balcanica Boev [2]
Lullula minor Kessler
Lullula minuscula Kessler
Lullula neogradensis Kessler & Hír
Lullula parva Kessler
Lullula slivnicensis Boev [2]
Melanocorypha donchevi Boev [2]
Melanocorypha minor Kessler
Melanocorypha serdicensis Boev [2]
Praealauda hevesensis Kessler & Hír
Artamidae
Kurrartapu johnnguyeni Nguyen et al.
Bombycillidae
Bombycilla brevia Kessler
Bombycilla hamori Kessler & Hír
Bombycilla kubinyii Kessler
Cardinalidae
Passerina sp. ‘Yepómera, Mexiko’
Certhiidae
Certhia immensa Kessler
Cettidae
Cettia janossyi Kessler
Cettia kalmani Kessler
Cinclidae
Cinclus gaspariki Kessler
Cinclus major Kessler & Hír
Cinclus minor Kessler
Cinclosomatidae
Cinclosoma elachum Nguyen, Archer & Hand
Climacteridae
Climacteris sp. ‘Riversleigh, Australien’
Cormobates sp. ‘Riversleigh, Australien’
Corvidae
Corvus annectens Shufeldt
Corvus bragai Paiva [8]
Corvus harkanyensis Kessler
Corvus hungaricus Lambrecht
Corvus pliocaenicus (Portis)
Corvus praecorax Depéret
Corvus shufeldti Sharpe
Henocitta brodkorbi Holman
Miocitta galbraethi Brodkorb
Miocorvus larteti (Milne-Edwards)
Miopica paradoxa Kurotschkin & Sobolew
Pica pica ssp. major Mourer-Chauviré
Pica mourerae Seguí
Pica praepica Boev [9]
Protocitta ajax Brodkorb
Protocitta dixi Brodkorb
Pyrrhocorax graculus ssp. vetus Kretzoi
Dasyornithidae
Dasyornis walterbolesi Nguyen
Emberizidae
Emberiza gaspariki Kessler
Emberiza media Kessler
Emberiza parva Kessler
Emberiza polgardiensis Kessler
Emberiza shaamarica (Zelenkov & Kurochkin) [4]
Pedinorhis stirpsarcana Olson & McKittrick
Plectrophenax veterior Kessler
Estrildidae
Estrildidae gen. & sp. ‘Riversleigh, Australien’
Eurylaimidae
Eurylaimidae gen. & sp. ‘Wintersdorf, Deutschland’
Wieslochia weissi Mayr & Manegold [9]
Fringillidae
Carduelis kretzoii Kessler
Carduelis lambrechti Kessler
Carduelis medius Kessler
Carduelis parvulus Kessler
Coccothraustes balcanicus Boev
Coccothraustes major Kessler
Coccothraustes simeonovi Boev
Fringilla kormosi Kessler
Fringilla petenyii Kessler
Loxia csarnotanus Kessler
Loxia patevi Boev
Pinicola kubinyii Kessler
Pyrrhula gali Kessler
Pyrrhula minor Kessler
Furnariidae
Pseudoseisuropsis nehuen Noriega
Pseudoseisuropsis cuelloi Claramunt & Rinderknecht
Pseudoseisuropsis wintu Stefanini et al.
Hirundinidae
Delichon major Kessler
Delichon polgardiensis Kessler
Delichon pusillus Kessler
Hirundinidae gen. & sp. ‘Langebaanweg, Südafrika’ (mehrere spp.)
Hirundo aprica Feduccia
Hirundo gracilis Kessler
Hirundo major Kessler
Riparia minor Kessler
Icteridae
Cremaster tytthus (Brodkorb)
Euphagus magnirostris (Miller)
Icterus turmalis Steadman & Oswald [7]
Molothrus resinosus Steadman & Oswald [7]
Pandanaris convexa (Miller)
Pyelorhamphus molothroides (Miller)
Laniidae
Lanius capeki Kessler
Lanius hungaricus Kessler
Lanius intermedius Kessler
Lanius major Kessler
Lanius schreteri Kessler & Hír
Leiotrichidae
Turdoides borealis Jánossy
Locustellidae
Locustella janossyi Kessler
Locustella kordosi Kessler
Locustella mana Kessler
Locustellidae gen. & sp. ‘Riversleigh, Australien’
Megalurus sp. ‘Riversleigh, Australien’
Maluridae
Maluridae gen. & sp. ‘Riversleigh, Australien’
Meliphagidae
Meliphagidae gen. & sp. ‘Riversleigh, Australien’ (mehrere spp.)
Menuridae
Menura tyawanoides Boles
Morsoravidae
Morsoravis sedilis Bertelli, Lindow, Dyke & Chiappe
Pumiliornis tessellatus Mayr
Sororavis solitarius Mayr & Kitchener
Motacillidae
Anthus antecedens Kessler & Hír
Anthus baranensis Kessler
Anthus hiri Kessler
Motacilla intermedia Kessler
Motacilla minor Kessler
Motacilla robusta Kessler
Muscicapidae
‘Emberiza’ pannonica Kessler
Erithacus horusitzkyi Kessler & Hír
Erithacus minor Kessler
Luscinia denesi Kessler
Luscinia pliocaenica Kessler
Luscinia praeluscinia Kessler & Hír
Monticola pongraczii Kessler
Muscicapa leganyii Kessler & Hír
Muscicapa miklosi Kessler
Muscicapa petenyii Kessler
Oenanthe kormosi Kessler
Oenanthe pongraczi Kessler
Phoenicurus baranensis Kessler
Phoenicurus erikai Kessler
Saxicola baranensis Kessler
Saxicola lambrechti Kessler
Saxicola magna Kessler
Saxicola parva Kessler
Namapsittidae
Namapsitta praeruptorum Mourer-Chauviré et al. [13]
Neosittidae
Daphoenositta trevorworthyi Nguyen
Oriolidae
Longimornis robustirostrata Boles
Oriolus beremendensis Kessler
Orthonychidae
Orthonyx kaldowinyeri Boles
Palaeoscinidae (?)
Palaeoscinis turdirostris Howard
Paridae
Parus medius Kessler
Parus parvulus Kessler
Parus robustus Kessler
Passerellidae
Ammodramus eurius Brodkorb
Ammodramus hatcheri (Shufeldt)
Pampaemberiza olrogi Agnolin
Passeridae
Passer hiri Kessler
Passer minusculus Kessler
Passer pannonicus Kessler
Passer predomesticus Tchernov
Petroicidae
Petroicidae gen. & sp. ‘Riversleigh, Australien’
Phylloscopidae
Phylloscopus miocaenicus Kessler & Hír
Phylloscopus pliocaenicus Kessler
Phylloscopus venczeli Kessler
Pomatostomidae
Pomatostomus sp. ‘Riversleigh, Australien’
Prunellidae
Prunella freudenthali Kessler
Prunella kormosi Kessler
Psittacopedidae
Eofringillirostrum boudreauxi Mayr, Ksepka & Grande
Eofringillirostrum parvulum Mayr, Ksepka & Grande
Parapsittacopes bergdahli Mayr [12]
Psittacomimus eos Mayr & Kitchener
Psittacopes lepidus Mayr & Daniels
Psittacopes occidentalis Mayr & Kitchener
Ptilonorhynchidae
Sericuloides marynguyenae Nguyen [14]
Regulidae
Regulus bulgaricus Boev
Regulus pliocaenicus Kessler
Sittidae
Sitta gracilis Kessler
Sitta ‘pusilla‘ Kessler (Artname bereits vergeben!)
Sitta senogalliensis Portis
Sitta villanyensis Kessler
Sturnidae
Dobrosturnus kardamensis Boev [5]
Sturnus baranensis Kessler
Sturnus brevis Kessler
Sturnus kretzoii Kessler & Hír
Sturnus pliocaenicus Kessler
Sylviidae
Sylvia intermedia Kessler
Sylvia pussila Kessler
Tichodromidae
Tichodroma apeki Kessler
Troglodytidae
Troglodytes robustus Kessler
Turdidae
Meridiocichla salotti Louchart
Turdicus minor Kessler & Hír
Turdicus pannonicus Kessler
Turdus major Kessler
Turdus medius Kessler
Turdus minor Kessler
Turdus miocaenicus Kessler
Turdus polgardiensis Kessler
Zygodactylidae
Eozygodactylus americanus Weidig
Primoscens carolinae Mayr & Kitchener
Primoscens minutus Harrison & Walker
Primozygodactylus ballmanni Mayr
Primozygodactylus danielsi Mayr
Primozygodactylus eunjooae Mayr & Zelenkov
Primozygodactylus longibrachium Mayr
Primozygodactylus major Mayr
Primozygodactylus quintus Mayr
Zygodactylus grandei Smith, DeBee & Clarke
Zygodactylus grivensis Ballmann
Zygodactylus ignotus Ballmann
Zygodactylus luberonensis Mayr
“Zygodactylus” ochlurus Hieronymus, Waugh & Clarke
*********************
Referenzen:
[1] Jeno Kessler; János Hír: The avifauna in North Hungary during the Miocene Part II. Földtani Közlöny 142(2): 149-168. 2012
[2] Zlatozar Boev: Neogene Larks (Aves: Alaudidae (Vigors, 1825)) from Bulgaria. Acta Zoologica Bulgarica 64(3): 295-318. 2012
[3] Nikita V. Zelenkov: The revised avian fauna of Rudabànya (Hungary, Late Miocene). Contribuciones del Museo Argentino de Ciencias Naturalis “Bernardino Rivadavia” 7: 253-266. 2017
[4] E. S. Palastrova; N. V. Zelenkov: A fossil bunting Emberiza shaamarica (Aves, Emberizidae) from the Upper Pliocene of Central Asia. Paleontological Journal 54: 652-661. 2020
[5] Zlatozar N. Boev: A new Middle Miocene starling (Sturnidae Rafinesque, 1815) from Karma (NE Bulgaria). Bulletin of the Natural History Museum – Plovdiv 5: 33-41. 2020
[6] Nicolas Dussex; David W. G. Stanton; Hanna Sigeman; Per G. P. Ericson; Jacquelyn Gill; Daniel C. Fisher; Albert V. Protopopov; Victoria L. Herridge; Valery Plotnikov; Bengt Hansson; Love Dalén: Biomolecular analyses reveal the age, sex and species identity of a near-intact Pleistocene bird carcass. Communications Biology 3: 1-6. 2020
[7] David W. Steadman; Jessica A. Oswald: New species of troupial (Icterus) and cowbird (Molothrus) from ice-age Peru. The Wilson Journal of Ornithology 132 (1): 91–103. 2020
[8] Marco Pavia: Palaeoenvironmental reconstruction of the Cradle of Humankind during the Plio-Pleistocene transition, inferred from the analysis of fossil birds from Member 2 of the hominin-bearing site of Kromdraai (Gauteng, South Africa) Quaternary Science Reviews. 248: Article 106532. doi:10.1016/j.quascirev.2020.106532. 2020
[9] Jon Fjeldså; Les Christidis; Per G. P. Ericson: The Largest Avian Radiation: The Evolution of Perching Birds, or the Order Passeriformes. Lynx Edicions 2020
[10] Z. N. Boev: An early Pleistocene magpie (Pica praepica sp. n.) (Corvidae Leach, 1820) from Bulgaria. Bulletin of the Natural History Museum – Plovdiv 6: 51–59. 2021
[11] Zbigniew M. Bochenski; Teresa Tomek; Małgorzata Bujoczek; Grzegorz Salwa: A new passeriform (Aves: Passeriformes) from the early Oligocene of Poland sheds light on the beginnings of Suboscines. Journal of Ornithology 2021
[12] Gerald Mayr: A remarkably complete skeleton from the London Clay provides insights into the morphology and diversity of early Eocene zygodactyl near-passerine birds. Journal of Systematic Palaeontology 12 Jan 2021
[13] Gerald Mayr: Paleogene Fossil Birds. 2nd ed. edition 2022
[14] Jacqueline M. T. Nguyen: The earliest record of bowerbirds (Passeriformes, Ptilonorhynchidae) from the Oligo-Miocene of northern Australia. Alcheringa: An Australasian Journal of Palaeontology, DOI: 10.1080/03115518.2023.2180537. 2023
*********************
bearbeitet: 09.04.2023
Aus Eins mach Zwei: Sängervireo
Der Sängervireo (Vireo gilvus (Vieillot)) ist eine mittelgroße, unauffällig gefärbte Vogelart, die in großen Teilen Nord- und im Norden Mittelamerikas brütet; der Art werden insgesamt vier Unterarten zugeordnet von denen die Nominatform das größte Verbreitungsgebiet aufweist.

Foto: Mark Dennis
https://www.inaturalist.org/people/md-in-ns15
https://creativecommons.org/licenses/by-nc/4.0/
*********************
Die restlichen drei Unterarten unterscheiden sich nicht zuletzt anhand ihres Gesangs von der Nominatform sondern vor allem auch anhand ihrer mitochondrialen DNA und sollten daher zusammen als eigenständige Art, Westlicher Sängervireo (Vireo swainsoni Baird), abgespalten werden.

Foto: Garth Harwood
https://www.inaturalist.org/people/garth_harwood
https://creativecommons.org/licenses/by/4.0/

Foto: Oscar Johnson
https://www.inaturalist.org/people/henicorhina
http://creativecommons.org/licenses/by-nc-nd/4.0/

Foto: Steven Mlodinow
https://www.inaturalist.org/people/mlodinow
https://creativecommons.org/licenses/by-nc/4.0/
*********************
Quelle:
[1] Scott F. Lovell; M. Ross Lein; Sean M. Rogers: Cryptic speciation in the Warbling Vireo (Vireo gilvus). Ornithology 139(3): 1-16. 2021
*********************
bearbeitet: 01.04.2023
Aus Eins mach Vier: Fuchsscheitelvireo
Der Fuchscheitelvireo (Tunchiornis ochraceiceps (Sclater)) ist eine der zahlreichen Vogelarten, über die so gut wie nichts bekannt ist, davon kann man sich leicht selbst überzeugen, wenn man einfach mal ein paar Blicke auf die verschiedenen Wikipedia-Einträge wirft, die es zu dieser Art gibt, diese stimmen nämlich nicht einmal im Gattungsnamen überein.
Die Art, oder besser der Artkomplex, bewohnt die bewaldeten Gebiete Mittelamerikas und des nördlichen Südamerikas.
Beschrieben wurde der Fuchsscheitelvireo ursprünglich als Mitglied der Gattung Hylophilus, deren Vertreter durchweg recht klein und eher unauffällig bräunlich gefärbt sind. Später stellte sich jedoch heraus, dass diese Gattung polyphyletisch ist, das bedeutet, dass ihre Vertreter, trotz ihres sehr ähnlichem Äußeren, keinen gemeinsamen Ursprung haben und somit eben auch nicht in dieselbe Gattung gestellt werden können und somit wurde für den Fuchsscheitelvireo eine neue Gattung erstellt. [1]
Von nun an wurde diese neue Gattung mal als monotypisch betrachtet und mal in zwei Arten aufgespalten, insgesamt wurden, je nach Autor, bis zu zehn oder gar mehr Taxa (Unterarten) anerkannt.
***
Anhand von genetischen Untersuchungen weiß man nun aber mittlerweile, dass sich einige der vermeintlichen Unterarten vor zum Teil 10 Millionen Jahren voneinander getrennt haben, was bedeutet, dass sie tatsächlich als eigenständige Arten betrachtet werden müssen, trotz äußerlicher Ähnlichkeiten, und dass andere überhaupt keine taxonomische Berechtigung haben. [2]
Nach dieser jüngsten Revision verbleiben die folgenden sechs Taxa.:
Rostscheitelvireo (Tunchiornis ferrugineifrons (Sclater))
Goldscheitelvireo (Tunchiornis luteifrons (Sclater))
Pulun-Fuchscheitelvireo (Tunchiornis ochraceiceps ssp. bulunensis (Hartert))
Fuchsscheitelvireo (Tunchiornis ochraceiceps ssp. ochraceiceps (Sclater))
Gelblicher Rotscheitelvireo (Tunchiornis rubrifrons ssp. lutescens (Snethlage))
Rotscheitelvireo (Tunchiornis rubrifrons ssp. rubrifrons (Sclater & Salvin))
*********************

Darstellung aus: ‘P. L. Sclater: On the genus Hylophilus. The Ibis 4(19): 293-311. 1881’
(public domain)
*********************
Quellen:
[1] David L. Slager; John Klicka: Polyphyly of Hylophilus and a new genus for the Tawny-crowned Greenlet (Aves: Passeriformes: Vireonidae). Zootaxa. 3884 (2): 194–196. 2014
[2] Nelson Buainain; Marina F. A. Maximiano; Mateus Ferreira; Alexandre Aleixo; Brant C. Faircloth; Robb T. Brumfield; Joel Cracraft; Camila C. Ribas: Multiple species and deep genomic divergences despite little phenotypic differentiation in an ancient Neotropical songbird, Tunchiornis ochraceiceps (Sclater, 1860) (Aves: Vireonidae). Molecular Phylogenetics and Evolution 162: 1-17. 2021
*********************
bearbeitet: 27.03.2023
How to paint realistic birds in watercolour ….
*********************
edited: 22.03.2023
Kleiber


*********************
bearbeitet: 19.03.2023
QM F20688 und QM F24685
Hinter diesen beiden Buchstaben- und Nummernfolgen verbergen sich zwei winzige Knochenbruchstücke, die aus dem frühen Eozän Australiens stammen und von den ältesten bislang bekannten echten Singvögeln stammen.
QM F20688 ist ein Fragment des proximalen Teils eines Carpometacarpus eines mittelgroßen -, QM F24685 wiederum ein Fragment des distalen Teils eines Tibiotarsus eines sehr kleinen Vogels.
Beide Knochenfragmente stammen wohl tatsächlich von echten Singvögeln, die, wie man mittlerweile weiß, in der australischen Region entstanden sind und sich von dort über die gesamte Welt ausgebreitet haben; im frühen Eozän mögen dies Verwandte oder Vertreter der so genannten Basalen Oscines gewesen sein, einer Gruppe von Singvögeln, die sich zu jener Zeit von den Passerines abspaltete und auch heute noch existiert aber ausschließlich in der australischen Region verbreitet ist.
*********************
Referenzen:
[1] Walter E. Boles: The world’s oldest songbird. Nature 374: 21-22. 1995
[2] Walter E. Boles: Fossil Songbirds (Passeriformes) from the Early Eocene of Australia. Emu 97(1): 43-50. 1997
[3] Alexandre Pedro Selvatti; Luiz Pedreira Gonzaga; Claudia Augusta de Moraes Russo: A Paleogene origin for crown passerines and the diversification of the Oscines in the New World. Molecular Phylogenetics and Evolution 88: 1-5. 2015
*********************
bearbeitet: 19.03.2023
Enantiornithes – die ‘anderen’ Vögel
Die Enantiornithes oder Gegenvögel, existierten, soweit bisher bekannt, ausschließlich während der Kreidezeit und zwar vom Beginn bis zum Ende dieser Periode, dies sind ca. 80 Millionen Jahre.
Diese erstaunlich erfolgreichen Vögel unterschieden sich von den heutigen anhand einiger Eigenarten.: So wuchsen die meisten Arten offenbar mehrere Jahre bis sie vollkommen ausgewachsen waren, konnten aber wohl schon kurz nach dem Schlupf fliegen, waren selbst als Jungvögel schon mit einem kompletten Erwachsenengefieder bestückt und konnten ebenso bereits im juvenilen Alter selbst Nachwuchs bekommen. Die Vögelchen besaßen meist Zähne in ihren Kiefern, hatten aber keine Schnäbel im eigentlichen Sinne; sie hatten oft, aber längst nicht immer, Krallenbewehrte Finger an den Händen; auch ihre Federn unterschieden sich von denen heutiger Vögel, vor allem die so genannten Schaftdominierten Federn (rachis dominated feathers), die viele Arten anstatt von Schwanzfedern besaßen; der Schaft dieser Federn war nicht zylindrisch sondern flach und bestand offenbar oft tatsächlich aus nur einer Zellschicht; außerdem scheinen diese Federn bei einigen Arten sogar Sollbruchstellen besessen zu haben um im Notfall einfach abgestoßen werden zu können. [1][2][3]
Einige Arten waren erstaunlich winzig, so dass sie selbst heutige Kolibris groß erscheinen lassen würden.
*********************
Referenzen:
[1] Lida Xing; Pierre Cockx; Ryan C. McKellar; Jingmai O’Connor: Ornamental feathers in Cretaceous Burmese amber: resolving the enigma of rachis-dominated feather structure. Journal of Palaeogeography 7(13): 1-18. 2018
[2] Nathan R.Carroll; Luis M. Chiappe; David J. Bottjer: Mid-cretaceous amber inclusions reveal morphogenesis of extinct rachis-dominated feathers. Scientific Reports 9: 1-9. 2019
[3] Shuo Wang; Wei-Ling Chang; Qiyue Zhang; Menglu Ma; Feng Yang; De Zhuo; Harn I-Chen Hans; Rui Yang; Ping Wu; Michael Habib; Wen-Tau Juan; Cheng-Ming Chuong: Variations of Mesozoic feathers: Insights from the morphogenesis of extant feather rachises. Evolution 74(9): 2121-2133. 2020
*********************
bearbeitet: 18.03.2023
Fossile Ordnung incertae sedis
Jinguofortisidae
Chongmingia zhengi Wang et al. [2]
Cratonavis zhui Li et al. [3]
Jinguofortis perplexus Wang et al. [1]
*********************
Referenzen:
[1] Min Wang; Xiaoli Wang; Yan Wang; Zhonghe Zhou: A new basal bird from China with implications for morphological diversity in early birds. Scientific Reports 6(1), 19700; doi: 10.1038/srep19700. 2016
[2] Min Wang; Thomas A. Stidham; Zhonghe Zhou: A new clade of basal Early Cretaceous pygostylian birds and developmental plasticity of the avian shoulder girdle. Proceedings of the National Academy of Sciences USA 115(42): 10708-10713. 2018
[3] Zhiheng Li; Min Wang; Thomas A. Stidham; Zhonghe Zhou: Decoupling the skull and skeleton in a Cretaceous bird with unique appendicular morphologies. Nature Ecology & Evolution 7: 20-31. 2023
*********************
bearbeitet: 17.03.2023
Fossile Ordnung incertae sedis
Eurofluvioviridavidae (?)
Eurofluvioviridavis robustipes Mayr [1]
Vastanavidae (?)
Avolatavis europaeus Mayr & Kitchener [2]
Avolatavis tenens Ksepka & Clarke [1]
Vastanavis cambayensis Mayr et al. [1]
Vastanavis eocaena Mayr et al. [1]
***
Die drei hier aufgeführten Gattungen scheinen näher miteinander verwandt gewesen zu sein als mit anderen Taxa, ob sie allerdings wirklich allesamt in eine gemeinsame Ordnung gestellt werden können, ist zweifelhaft.
*********************
Referenzen:
[1] Gerald Mayr: Paleogene Fossil Birds. 2nd ed. edition 2022
[2] Gerald Mayr; Andrew C. Kitchener: The Vastanavidae and Messelasturidae (Aves) from the early Eocene London Clay of Walton-on-the-Naze (Essex, UK). Neues Jahrbuch für Geologie und Paläontologie 307(2): 113-139. 2023
*********************
bearbeitet: 17.03.2023
Fossile Sapeornithiformes
Sapeornithidae
Sapeornis chaoyangensis Zhou & Zhang
*********************
Quelle:
[1] Z. Zhou; F. Zhang: Anatomy of the primitive bird Sapeornis chaoyangensis from the Early Cretaceous of Liaoning, China. Canadian Journal of Earth Sciences 40: 731-747. 2003
*********************
bearbeitet: 16.03.2023
Fossile Jeholornithiformes
Jeholornithidae (?)
Jeholornis curvipes Lefèvre et al.
Jeholornis palmapenis O’Connor et al.
Jeholornis prima Zhou & Zhang
Jixiangornis orientalis Ji et al.
Kompsornis longicaudus Wang, Huang, Kundrát, Cau, Liu, Wang & Juae [1]
Neimengornis rectusmim Wang et al.
Shenzouraptor sinensis Ji et al.
*********************
Referenzen:
[1] Xuri Wang; Jiandong Huang; Martin Kundrát; Andrea Cau; Xiaoyu Liu; Yang Wang; Shubin Juae: A new jeholornithiform exhibits the earliest appearance of the fused sternum and pelvis in the evolution of avialan dinosaurs. Journal of Asian Earth Sciences 2020
*********************
bearbeitet: 14.03.2023
Morsoravis sedilis Bertelli, Lindow, Dyke & Chiappe

*********************
bearbeitet: 08.03.2023
Sororavis solitarius Mayr & Kitchener

*********************
bearbeitet: 08.03.2023
Cinclidae – Wasseramseln
Diese kleine Familie umfasst fünf Arten, von denen drei auf der Nordhalbkugel – sowie zwei in Südamerika verbreitet sind; die drei Arten der Nordhalbkugel sind die einzigen wirklich aquatisch lebenden Singvögel der Welt, die beiden südamerikanischen Arten (Weißkehl-Wasseramsel (Cinclus leucocephalus Tschudi) und Rostkehl-Wasseramsel (Cinclus schulzii Cabanis)) leben zwar ebenfalls an Gewässern, scheinen eher weniger an ein Wasserleben angepasst zu sein.
Die Nordhalbkugel-Vögel leben an und in schnellfließenden Gewässern und suchen ihre Nahrung aktiv tauchend, wobei sie sich unter Wasser mit den Flügel schlagend vorwärtsbewegen, tatsächlich ganz ähnlich wie z.B. Alken (Alcidae) und Pinguine (Spheniscidae). Sie zeigen daher auch einige konvergente Anpassungen an diese Lebensweise, die sie mit anderen tauchenden Vogelformen teilen, wie z.B. auf schmale Schlitze reduzierte Nasenöffnungen, verkürzte Fußknochen oder leicht verdickte Knochenwände (verglichen mit anderen Singvögeln).
***
Die Darstellung zeigt zwei Unterarten der Eurasischen Wasseramsel (Cinclus cinclus (L.)) sowie die einfarbig dunkelbraun gefärbte Pallaswasseramsel (Cinclus pallsii Temminck) aus Ostasien.
*********************

(public domain)
*********************
Quelle:
[1] N. Adam Smith; Krista L. Koeller; Julia A. Clarke; Daniel T. Ksepka; Jonathan S. Mitchell; Ali Nabavizadeh; Ryan C. Ridgley; Lawrence M. Witmer: Convergent evolution in dippers (Cinclidae): the only wing-propelled diving songbirds. The Anatomical Record 1-29. 2021
*********************
bearbeitet: 07.03.2023
Elefantöse Neuigkeiten!
Seit der letzten Revision der Familie der so genannten Elefantenvögel (Aepyornithidae) [1] war deren Artenzahl von sieben auf vier geschrumpft.:
Aepyornithidae
Aepyornis hildebrandti Burckhardt
Aepyornis maximus Geoffroy Saint Hilaire
Mullerornis modestus (Milne-Edwards & Grandidier)
Vorombe titan (Andrews)
***
Eine ganz neue Studie [2], in der ca. 1000 Jahre alte Eierschalen untersucht wurden, zeigt nun, dass einige dieser Arten eher weitläufig miteinander verwandt waren, so dass man sie sogar in verschiedene Familien stellen kann (ähnlich wie bei den neuseeländischen Moa-Arten, die auf drei Familien aufgeteilt werden).
Mullerornis modestus (Milne-Edwards & Grandidier) ist nun somit die einzige (derzeit anerkannte) Art der neuen Familie Mullerornithidae.
Einiges deutet auch darauf hin, dass Aepyornis maximus Geoffroy Saint Hilaire und Vorombe titan (Andrews) ein und dieselbe Art darstellen, dann würde es sich hierbei um einen der extremsten Fälle von Geschlechtsdimorphismus handeln, den man aus dem Vogelreich kennt, wahrscheinlich waren nämlich, wie auch bei anderen Ratiten, die Weibchen größer als die Männchen, in diesem Fall nur eben sehr viel größer (einen ähnlich extremen Geschlechtsdimorphismus findet man aber auch bei einigen der neuseeländischen Moa-Arten).
Zum Schluss ergab die Studie auch noch, dass es im äußersten Norden Madagaskars eine genetisch isolierte Population der Gattung Aepyornis gab, bei der es sich um eine eigenständige Art gehandelt haben mag.
***
Und so sieht die aktualisierte Nomenklatur der Elefantenvögel nun aus.:
Aepyornithidae
Aepyornis hildebrandti Burckhardt
Aepyornis maximus Geoffroy Saint Hilaire
Aepyornis sp. ‘Irodo’
Mullerornithidae
Mullerornis modestus (Milne-Edwards & Grandidier)
*********************
Quellen:
[1] James P. Hansford; Samuel T. Turvey: Unexpected diversity within the extinct elephant birds (Aves: Aepyornithidae) and a new identity for the world’s largest birds. Royal Society Open Science 5(9): 1-28. 2018
[2] Alicia Grealy; Gifford H. Miller; Matthew J. Phillips; Simon J. Clarke; Marilyn Fogel; Diana Patalwala; Paul Rigby; Alysia Hubbard; Beatrice Demarchi; Matthew Collins; Meaghan Mackie; Jorune Sakalauskaite; Josefin Stiller; Julia A. Clarke; Lucas J. Legendre; Kristina Douglass; James Hansford; James Haile; Michael Bunce: Molecular exploration of fossil eggshell uncovers hidden lineage of giant extinct bird. Nature Communications 14(914): 1-14. 2023
*********************
bearbeitet: 07.03.2023
Fossile Pengornithiformes (?)
Pengornithidae
Chiappeavis magnapremaxillo O’Connor et al. = Pengornis houi Zhou, Clarke, & Zhang? [1]
Eopengornis martini Wang et al.
Parapengornis eurycaudatus Hu et. al. = Pengornis houi Zhou, Clarke, & Zhang? [1]
Pengornis houi Zhou, Clarke, & Zhang
Yuanchuavis kompsosoura Wang et al. = Pengornis houi Zhou, Clarke, & Zhang? [2][3]
*********************
Quellen:
[1] Mickey Mortimer: Chiappeavis is just another Pengornis. The Theropod Database Blog, January 6, 2016
[2] Min Wang; Jingmai O’Connor; Thao Zhao; Yangon Pan; Xiaoting Zheng; Xioli Wang; Zhonghe Zhou: An Early Cretaceous enantiornithine bird with a pintail. Current Biology 31(21): 4845-4852. 2021
[3] Mickey Mortimer: Neimengornis the chimaera, “Yuanchuavis” is another Pengornis and more – October 2021 Database update. The Theropod Database Blog, November 8, 2021
*********************
bearbeitet: 07.03.2023
***
Hinweis: Diese Ordnung ist vermutlich nicht gültig.
Fossile Gaviiformes
Gaviidae
Colymboides anglicus Lydekker
Colymboides belgicus Mayr & Smith
Colymboides metzleri Mayr
Colymboides minutus Milne-Edwards
Gavia egeriana Švec
Gaviella pusilla (Shufeldt)
Nasidytes ypresianus Mayr & Kitchener [1]
*********************
Quellen:
[1] Gerald Mayr; Markus Poschmann: A loon leg (Aves, Gaviidae) with crocodilian tooth from the Late Oligocene of Germany. Waterbirds 32(3): 468-471. 2009
[2] Gerald Mayr; Andrew C. Kitchener: Oldest fossil loon documents a pronounced ecomorphological shift in the evolution of gaviiform birds. Zoological Journal of the Linnean Society. 2022
*********************
bearbeitet: 07.03.2023
Fossile Galliformes
Familie incertae sedis
Archaealectrornis sibleyi Crowe & Short
Archaeophasianus mioceanus Lambrecht
Archaeophasianus roberti (Stone)
Argillipes aurorum Harrison & Walker
Argillipes paralectoris Harrison & Walker
Austinornis lentus Marsh
Bumbanipodius magnus Zelenkov [3]
Bumbanortyx transitoria Zelenkov [3]
Chambiortyx cristata Mourer-Chauviré
Coturnipes cooperi Harrison & Walker
Namaortyx sperrgebietensis Mourer-Chauviré
Palaeonossax senectus Wetmore
Palaeorallus alienus Kuročkin
Sobniogallus albinojamrozi Tomek et al.
Procrax brevipes Tordoff & Macdonald
Tegulavis corbalani Mourer-Chauviré et al.
Cracidae
Boreortalis laesslei Brodkorb
Ortalis affinis Feduccia & Wilson
Gallinuloididae
Gallinuloides wyomingensis Eastman
Paraortygoides messelensis Mayr
Paraortygoides radagasti Dyke & Gulas
Megapodidae
Garrdimalga mcnamarai Shute, Prideaux & Worthy
Latagallina naracoortensis Shute, Prideaux & Worthy
Latagallina olsoni Shute, Prideaux & Worthy
Progura campestris Shute, Prideaux & Worthy
Progura gallinacea De Vis
Ngawupodius minya Boles & Ivison
Numididae
Telecrex grangeri Wetmore
Odontophoridae
Cyrtonyx tedfordi Miller
Miortyx aldeni Howard
Miortyx teres Miller
Nanortyx inexpectatus Weigel
Neortyx peninsularis Holman
Paraortygidae
Paraortyx brancoi Gaillard
Paraortyx lorteti Gaillard
Pirortyx major (Gaillard)
Scopelortyx klinghardtensis Mourer-Chauvire et al.
Taoperdix miocaena Ballman
Taoperdix pessieti Gervais
Xorazmortyx turkestanensis Zelenkov & Panteleyev
Phasianidae
Alectoris baryosefi Černov
Alectoris peii Hou
“Alectoris” pliocaena Tugarinov
Bantamyx georgicus Kurochkin
Bonasa praebonasia (Jánossy)
Centuriavis lioae Ksepka et al.
Chauvireria bulgarica Boev [1]
Dendragapus gilli (Shufeldt)
Dendragapus lucasi Howard
Dendragapus nanus (Shufeldt)
Diangallus mious Hou
Eurobambusicola turolicus Zelenkov
“Gallus” aesculapii Jánossy
“Gallus” beremendensis Jánossy
“Gallus” europaeus Harrison
Gallus georgicus Burčak-Abramovič & Potapova
Gallus imereticus Burčak-Abramovič & Potapova
Gallus karabachensis Baryšnikov & Potapova
Gallus kudarensis Burčak-Abramovič & Potapova
Gallus meschtscheriensis Burčak-Abramovič & Potapova
Gallus sp. ‘Trinka Cave, Moldau’
Gallus sp. ‘Krivtcha Cave, Ukraine’
Gallus tamanensis Burčak-Abramovič & Potapova
Lagopus balcanicus Boev
Lagopus lagopus ssp. noaillensis Mourer-Chauviré
Lagopus mutus ssp. correzensis Mourer-Chauviré
Linquornis gigantis Yeh
Lophogallus naranbulakensis Zelenkov & Kurochkin
Megalocoturnix cordoni Sánchez Marco
Meleagris altus Marsh
Meleagris californica (Miller)
Meleagris celer Marsh
Meleagris leopoldi Miller & Bowman
Meleagris richmondi Shufeldt
Meleagris tridens Wetmore
Miophasianus altus Milne-Edwards
Miophasianus desnoyersi Milne-Edwards
Miophasianus medius Milne-Edwards
Mioryaba magyarica Zelenkov
Palaeocryptonyx depereti Gaillard
Palaeocryptonyx donnezani Deperet
Palaeocryptonyx gaillardi Ennouchi
Palaeocryptonyx grivensis Ennouchi
Palaeoperdix longipes Milne-Edwards
Palaeoperdix prisca Milne-Edwards
Palaeoperdix sansaniensis Milne-Edwards
Palaeortyx blanchardi Milne-Edwards
Palaeortyx brevipes Milne-Edwards
Palaeortyx caluxyensis Lydekker
Palaeortyx depereti Ennouchi
Palaeortyx edwardsi Depéret
Palaeortyx gaillardi Lambrecht
Palaeortyx gallica Milne-Edwards
Palaeortyx grivensis Lydekker
Palaeortyx intermedia Ballman
Palaeortyx joleaudi Ennouchi
Palaeortyx major Gaillard
Palaeortyx maxima Lydekker
Palaeortyx media Milne-Edwards
Palaeortyx miocaena Gaillard
Palaeortyx ocyptera Milne-Edwards
Palaeortyx phasianoides Milne-Edwards
Palaeortyx volans Gohlich & Pavia
Panraogallus hezhengensis Li et. al.
Paralyra atavus (Jánossy)
Pavo bravardi (Gervais)
Pavo moldovicus (Bocheński & Kurochkin)
Pavo sp. ‘Aramis, Ethiopia’
Pediocetes lucasi Shufeldt
Pediocetes nanus Shufeldt
Perdix palaeoperdix Mourer-Chauviré
Phasianus bulgaricus Boev [2]
Pliogallus csarnotanus Kessler & Horváth [4]
Plioperdix hungarica Jánossy
Proagriocharis kimballensis Martin & Tate
Rhegminornis calobates Wetmore
Rustaviornis georgicus Burchak-Abramovich & Meladze
Schaubortyx keltica Eastman
Shandongornis shanwangensis Yeh
Shandongornis yinanensis Yeh
Shanxiornis fenyinis Wang et al.
Syrmaticus phasianoides (Jánossy)
Tetrao conjugens Jánossy
Tetrao macropus Jánossy
Tetrao partium (Kretzoi)
Tetrao praeurogallus Jánossy
Tetrao rhodopensis Boev
Titanoperdix felix Zelenkov
Tologuica aurorae Zelenkov & Kurochkin
Tologuica karhui Zelenkov & Kurochkin
“Tympanuchus” lulli Shufeldt
“Tympanuchus” stirtoni Miller
Quercymegapodiidae
Ameripodius alexis Mourer-Chauviré
Ameripodius silvasantosi Alvarenga
Ludiortyx hoffmanni (Gervais)
Quercymegapodius brodkorbi Mourer-Chauviré
Quercymegapodius depereti (Gaillard)
Taubacrex granivora Alvarenga
***
Beachte, dass diese Liste nicht komplett ist.
*********************
Quellen:
[1] Zlatozae Boev: Chauvireria bulgarica sp. n. — an extinct Early Pleistocene small phasianid of Phasianinae Horsfield, 1821 from Bulgaria. Historia naturalis bulgarica 41(8): 55-70. 2020
[2] Zlatozar Boev: First European Neogene record of true pheasants from Gorna Sushitsa (SW Bulgaria). Historia naturalis bulgarica 41(5): 33-39. 2020
[3] N. V. Zelenkov: New bird taxa (Aves: Galliformes, Gruiformes) from the early Eocene of Mogolia. Paleontological Journal 545(4). 2021
[4] E. Kessler; I. Horváth: Presentation of so far undetermined bird remains from the Pliocene of Beremend 26 and Csarnóta 2 and 4 (Baranya county, South Hungary). Ornis Hungarica 30: 47–68. 2022
*********************
bearbeitet: 18.04.2024
Fossile Confuciusornithiformes
Confuciusornithidae
Changchengornis hengdaoziensis Ji, Chiappe & Ji
Confuciusornis dui Hou et al.
Confuciusornis sanctus Hou et al.
Confuciusornis shifan Wang et al.
Eoconfuciusornis zhengi Zhang, Zhou & Benton
Yangavis confucii Wang & Zhou
*********************
bearbeitet: 23.12.2022
Fossile Leptosomiformes
Familie incertae sedis
Lapillavis incubarens Mayr (?)
Lutavis platypelvis Mayr & Kitchener
Leptosomidae
Plesiocathartes europaeus Gaillard
Plesiocathartes geiselensis Mayr
Plesiocathartes insolitipes Mayr & Kitchener
Plesiocathartes kelleri Mayr
Plesiocathartes major Weidig
Plesiocathartes (?) sp. ‘Egem, Belgium’
Plesiocathartes (?) sp. ‘India’
Plesiocathartes sp. ‘London Clay’
Plesiocathartes (?) sp. ‘Rivecourt-Petit Pâtis, France’
Plesiocathartes wyomingensis Weidig
Waltonavis danielsi Mayr & Kitchener
Waltonavis paraleptosomus Mayr & Kitchener
*********************
bearbeitet: 02.12.2022
Fossile Ichthyornithiformes
Ichthyornithidae
Ichthyornis dispar Marsh
Janavis finalidens Benito, Kuo, Widrig, Jagt & Field
*********************
bearbeitet: 01.12.2022
Brevirostruavis macrohyoideus Li et al.

*********************
bearbeitet: 16.11.2022
Seltsame Kreidezeit-Füße – Teil 2: DIP-V-15105a/b
DIP-V-15105a und DIP-V-15105b sind zwei Bernsteinfossilien, einmal ein fast vollständig erhaltener Fuß und zum anderen Teile eines Flügels, bzw. dessen Federn, beide gehören ziemlich wahrscheinlich zueinander.
DIP-V-15105a/b erreichte die Größe eines Kolibris, genauer gesagt eines winzigen Kolibris.
Der Fuß (es ist der rechte Fuß) ist interessanterweise bis fast zu den Zehen befiedert, wobei sich hier sogar zwei verschiedene Federformen finden, etwas längere, offenbar bräunlich gefärbte, dicht stehende Federn auf der Fußoberseite sowie vereinzelte, winzige borstenartige Federchen auf den eigentlichen Zehen selbst.
Dem Fußbau nach zu urteilen war dieser Vogel einem heutigen Baumläufer oder Kleiber vergleichbar, lebte also in den Wipfeln der Bäume und hielt sich bevorzugt an den größeren Ästen und den Stämmen auf wo er auf der Suche nach Insektenbeute schließlich mit dem Fuß in ausgetretenem Baumharz kleben blieb und so einen grausigen Tod fand.
*********************
Referenzen:
[1] Lida Xing; Ryan C. McKellar; Jingmai K. O’Connor; Ming Bai; Kuowei Tseng; Luis M. Chiappe: A fully feathered enantiornithine foot and wing fragment preserved in mid-Cretaceous Burmese amber. Scientific Reports 9(129): 1-9. 2019


leider habe ich gerade kein Gummibärchen zur Hand ….

*********************
bearbeitet: 15.11.2022
Farbtafel
*********************
bearbeitet: 03.11.2022
Neu beschrieben – Príncipe-Ohreule
Príncipe-Ohreule (Otus bikegila Melo et al.)
Diese Art von der Insel Príncipe, São Tomé und Príncipe, wurde offiziell im Jahr 2016 entdeckt, ist aber offenbar bereits 1928 in Aufzeichnungen von Ornithologen erwähnt worden; sie wurde jetzt wissenschaftlich beschrieben.
*********************

https://creativecommons.org/licenses/by/4.0/
*********************
Quelle:
[1] Martim Melo; Bárbara Freitas; Philippe Verbelen; Sátiro R. da Costa; Hugo Pereira; Jérôme Fuchs; George Sangster; Marco N. Correia; Ricardo F. de Lima; Angelica Crottini: A new species of scops-owl (Aves, Strigiformes, Strigidae, Otus) from Príncipe Island (Gulf of Guinea, Africa) and novel insights into the systematic affinities within Otus. ZooKeys 1126: 1-54. 2022
*********************
bearbeitet: 01.11.2022
Grünschlüpfer – genetische Untersuchungen decken eine zuvor unbekannte, versteckte Vielfalt auf
Der Grünschlüpfer ist mit ca. 8 cm Neuseelands kleinster Vogel und zugleich einer der urtümlichsten.
Derzeit werden dieser Art zwei Unterarten zugeordnet, die Nominatform, die auf der Südinsel lebt und die ssp. granti Mathews & Iredale (siehe Foto), die auf der Nordinsel vorkommt.

https://www.inaturalist.org/people/kevin_frank
https://creativecommons.org/licenses/by-nc/4.0/
*********************
Auf der Nordinsel weist die Art eine lückenhafte Verbreitung mit mehreren voneinander getrennten Populationen auf und lange Zeit wurde angenommen, dass dies durch Veränderungen der Umwelt durch menschliche Einflüsse hervorgerufen wurde – dem scheint aber nicht so zu sein. Eine Studie aus dem Jahr 2021 hat ergeben, dass es auf der Nordinsel mindestens drei genetisch voneinander unterscheidbare Populationen der Art gibt, von denen eine seit ca. 5 (südöstliche Küstenregion), eine weitere seit 2,5 bis 1,2 Millionen Jahren (Hauturu) genetisch isoliert sind. [1]
***
Es ist durchaus möglich, dass manche dieser genetisch isolierten Populationen den Status eigenständiger Unterarten zugeschrieben bekommen könnten, dafür sind aber weitere Untersuchungen notwendig, die auch die genetischen Daten der Nominatform von der Südinsel mit berücksichtigen.
*********************
Quelle:
[1] Sarah J. Withers; Stuart Parsons; Mark E. Hauber; Alistair Kendrick; Shane D. Lavery: Genetic divergence between isolated populations of the North Island New Zealand Rifleman (Acanthisitta chloris granti) implicates ancient biogeographic impacts rather than recent habitat fragmentation. Ecology and Evolution 2021; 00:1–17. https://doi.org/10.1002/ece3.7358
*********************
bearbeitet: 26.10.2022
Hybridkrähe

*********************
bearbeitet: 02.10.2022
Feder

*********************
bearbeitet: 02.10.2022
Feder

*********************
bearbeitet: 24.09.2022
Feder

*********************
bearbeitet: 05.06.2022
Messelirrisor parvus Mayr

*********************
bearbeitet: 30.05.2022
Primozygodactylus major Mayr
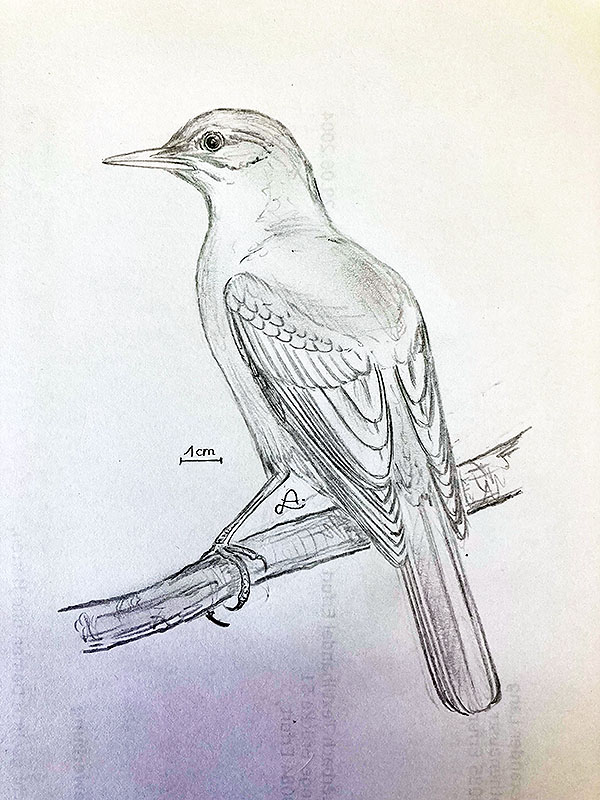
*********************
bearbeitet: 27.05.2022
Eurofluvioviridavis robustipes Mayr
*********************
bearbeitet: 27.05.2022
Primozygodactylus eunjooae Mayr & Zelenkov

*********************
bearbeitet: 24.05.2022
“Zygodactylus” ochlurus Hieronymus, Waugh & Clarke

*********************
bearbeitet: 24.05.2022
Fossile Ordnung incertae sedis
Eogruidae
Amphipelargus cracrafti (Harrison & Walker)
Amphipelargus majori Lydekker
Amphipelargus sp. ‘Kryopigi, Greece’
Eogrus aeola Wetmore
Eogrus crudus Kurochkin
Eogrus turanicus (Bendukidze)
Ergilornis rapidus Kozlova
Ergilornis minor (Kozlova)
Sinoergilornis guangheensis Musser, Li & Clarke [1]
Sonogrus gregalis Kurochkin
Urmiornis brodkorbi Karhu
Urmiornis dzabghanensis (Kurochkin)
Urmiornis maraghanus Mecquenem
Urmiornis orientalis (Kurochkin)
Urmiornis ukrainus Kurochkin
***
Es handelt sich hierbei um Vertreter der Palaeognathae und nicht, wie lange angenommen, um Kranichverwandte; die meisten Arten sind ausschließlich anhand von Fußknochen bekannt. [2][3]
*********************
Quellen:
[1] Grace Musser; Zhiheng Li; Julia A. Clarke: A new species of Eogruidae (Aves: Gruiformes) from the Miocene of the Linxia Basin, Gansu, China: Evolutionary and climatic implications. The Auk 137: 1-13. 2019
[2] G. Mayr; N. Zelenkov: Extinct crane-like birds (Eogruidae and Ergilornithidae) from the Cenozoic of Central Asia are indeed ostrich precursors. Ornithology 138(4): 2021
[3] Gerald Mayr: Paleogene Fossil Birds. 2nd ed. edition 2022
*********************
bearbeitet: 24.05.2022
Eremopezus eocaenus Andrews

*********************
edited: 23.05.2022
Remiornis heberti Lemoine
Beschrieben bereits 1881 aber bis heute nahezu unbekannt, ist diese Art, soweit mir bekannt, nur anhand von Bruchstücken des Schnabels sowie einiger Wirbel und eines Fußknochens bekannt.
Der Vogel war, laut einiger Autoren, zu Lebzeiten etwa so groß wie ein Emu (Dromaius novaehollandiae (Latham)) und mag ca. 55 kg gewogen haben [3]; ich persönlich komme beim Umrechnen aber nur auf eine Rückenhöhe von etwas über 70 cm.
Die Art ist bisher nur aus Frankreich bekannt und zwar aus Ablagerungen des Oberen Paläozän, also Schichten mit einem Alter von etwa 59 bis 56 Millionen Jahren, sie scheint außerdem mit keinem anderen Ratiten, lebend oder ausgestorben, näher verwandt gewesen zu sein.
*********************

*********************
Quellen:
[1] Victor Lemoine: Recherches sur les oiseaux fossiles des terrains tertiaires inférieurs des environs de Reims. Reims, Impr. F. Keller 1878-1881
[2] Eric Buffetaut; Delphine Angst: Stratigraphic distribution of large flightless birds in the Palaeogene of Europe and its palaeobiological and palaeogeographical implications. Earth-Science Reviews 138: 394-408. 2014
[3] Eric Buffetaut; Gaël de Ploëg: Giant birds from the uppermost Paleocene of Rivecourt (Oise, northern France). Boletim do Centro Português de Geo-História e Pré-História 2(1): 29-33. 2020
[4] Gerald Mayr: Paleogene Fossil Birds. 2nd ed. edition 2022
*********************
bearbeitet: 22.05.2022
Fossile Arten der Caprimulgiformes
Famile incertae sedis
Palaeopsittacus georgei Harrison
Ueekenkcoracias tambussiae Degrange, Diego Pol, Pablo Puerta & Peter Wilf [2]
Archaeotrogonidae
Archaeodromus anglicus Mayr [1]
Archaeotrogon cayluxensis Gaillard
Archaeotrogon hoffstetteri Mourer-Chauviré
Archaeotrogon nocturnus Mlíkovský
Archaeotrogon venustus Milne-Edwards
Archaeotrogon zitteli Gaillard
Hassiavis laticauda Mayr
Caprimulgidae
Ventivorus ragei Mourer-Chauviré (?)
*********************
Quellen:
[1] G. Mayr: An early Eocene fossil from the British London Clay elucidates the evolutionary history of the enigmatic Archaeotrogonidae (Aves, Strisores). Papers in Palaeontology advance online publication. doi: 10.1002/spp2.1392. 2021
[2] Federico J. Degrange; Diego Pol; Pablo Puerta; Peter Wilf: Unexpected larger distribution of paleogene stem-rollers (AVES, CORACII): new evidence from the Eocene of Patagonia, Argentina. Scientific Reports 11: 1363. 2021
[3] Gerald Mayr: Paleogene Fossil Birds. 2nd ed. edition 2022
*********************
bearbeitet: 24.04.2024
Messelornis cristata Hesse

*********************
bearbeitet: 21.05.2022
Palaeotis weigelti Lambrecht

*********************
bearbeitet: 21.05.2022
Parargornis messelensis Mayr

*********************
bearbeitet: 17.05.2022
Landbirds of the Brazilian Atlantic islands
In the Atlantic Ocean are three groups of islands that are politically part of Brazil and about which little seems to be known: the Fernando de Noronha Archipelago and the Rocks of Saint Peter and Saint Paul, both located near the equator and Trindade & Martim Vaz near the southern 20th parallel.
Interestingly, as many as four land bird species are known from these archipelagos, of which at least three are truly native and endemic: the Noronha Olive Tyrant (Elaenia ridleyana Sharpe) (Tyrannidae), the extinct Noronha Rail (cf. Rallus sp.) (Rallidae), the Noronha Vireo (Vireo gracilirostris Sharpe) (Vireonidae) and the Noronha Eared Dove (Zenaida auriculata ssp. noronha Sharpe) (Columbidae), all native to the largest archipelago, Fernando de Noronha.
***
There are also two more or less hypothetical forms for the second largest archipelago, Trindade & Martim Vaz: a pigeon form mentioned only by one of the first visitors to Trindade island at the end of the 17th century:
“In 1698 Dr. Halley visited the island, and says he found nothing living but doves and land-crabs.” [1]
And, for the time being, purely hypothetical but very probable, a rail, also for this one island.
***
No serious excavation appears to have taken place on any of the islands, such excavation would almost certainly unearth other bird forms that are now extinct.
*********************
References:
[1] R. Davis: Real Soldiers of Fortune. Charles Scribner’s Sons, New York 1906
[2] Robert Cushman Murphy: The birdlife of Trinidad Islet. The Auk 32(3): 332-348. 1915
[3] S. L. Olson: Natural history of vertebrates on the Brazilian islands of the mid South Atlantic. National Geographic Society Research Reports 13: 481-492. 1981
[4] Ruy José Válka Alves; Nílber Gonçalves da Silva: Três Séculos de História Natural na Ilha da Trindade com Comentários Sobre Sua Conservação. Smashwords 2016
*********************
edited: 05.07.2023
Pseudowissenschaftlicher Unsinn
Ich hatte mir eigentlich geschworen, nie wieder D. P.s Pterosaur Heresies-Webseite zu besuchen, aber nun ja, mir war langweilig ….
*********************
Am 26. Januar 2022 schreibt er etwas über den Blaubrustpipra.:

Foto: Dario Sanches
https://creativecommons.org/licenses/by-sa/2.0/
***
“Chrioxiphia [sic], the blue manakin, enters the LRT with its cuckoo relatives“
Übersetzung:
“Chrioxiphia, der Blaubrustpipra, betritt den LRT mit seinen Kuckucksverwandten“
***
“Chrioxiphia [sic] caudata (Shaw and Nodder 1793; 15cm) is the blue manakin, native to the Amazon. Many of the skull bones are fused. Phylogenetically (Fig. 2) manakins and their relatives are neotonous descendants of larger, long-legged ancestors like the South American seriema (Cariama) and its sister the flamingo (Phoenicopterus).“
Übersetzung:
“Chrioxiphia caudata (Shaw und Nodder 1793; 15 cm) ist der Blaubrustpipra, der im Amazonas beheimatet ist. Viele der Schädelknochen sind verschmolzen. Phylogenetisch (Abb. 2) sind Manakins und ihre Verwandten neotone Nachkommen größerer, langbeiniger Vorfahren wie der südamerikanischen Seriema (Cariama) und ihrer Schwester, dem Flamingo (Phoenicopterus).“
Uff, okay.
Nun, erst einmal lautet der Gattungsname der Art Chiroxiphia und nicht Chrioxiphia ….
Verschmolzene Schädelknochen stellen nicht wirklich irgendein relevantes Verwandtschaftsmerkmal dar, Vögel haben tatsächlich sehr viele verschmolzene Knochen, z.B. einige der Bein- oder Fußknochen. Pipras und Leierschwänze gehören innerhalb der Passeriformes zu je verschiedenen Unterordnungen, nämlich den Tyranni und den Passeri, sie sind damit in etwa so nah miteinander verwandt wie z.B. die Spechte mit den Tukanen. Seriemas und Flamingos sind nicht näher miteinander verwandt und stammen von unterschiedlichen Vorfahren ab, die sich schon sehr früh in der Stammesgeschichte der Vögel voneinander getrennt haben, vermutlich schon im Paläozän.
***
“Jungle-dwelling Chrioxiphia [sic] is related to jungle-dwelling Menura (the lyrebird) from Australia and jungle-dwelling Musophaga from Africa, these showy, colorful birds probably split phylogenetically prior to the Early Cretaceous appearance of the Atlantic Ocean. Few cuculids are long distance flyers. The showy birds in this clade prefer their jungles to crossing oceans.“
Übersetzung:
“Dschungelbewohnender Chrioxiphia ist mit dem im Dschungel lebenden Menura (dem Leierschwanz) aus Australien und dem im Dschungel lebenden Musophaga aus Afrika verwandt, diese auffälligen, farbenfrohen Vögel teilten sich wahrscheinlich phylogenetisch vor dem Erscheinen des Atlantischen Ozeans in der frühen Kreidezeit. Nur wenige Cuculiden sind Langstreckenflieger. Die auffälligen Vögel dieser Gruppe ziehen ihre Dschungel dem Überqueren von Ozeanen vor.“
Das verschiedene Vogelformen Dschungel bewohnen sagt rein gar nichts über ihre Verwandtschaft aus. Die Gattung Musophaga gehört zu den Turakos, einer ebenfalls recht alten Vogelordnung, die nicht näher mit den Passeriformes verwandt ist; nur weil all diese Vögel bunt gefärbt sind und Wälder bewohnen, macht sie dies nicht zu Verwandten! Diese Vogelordnungen haben sich auch nicht in der frühen Kreidezeit voneinander getrennt, das würde nämlich bedeuten, dass sie oder ihre Vorfahren bereits im Jura existiert haben müssten, dem ist aber nicht so, zu jener Zeit gab es gerade einmal die allerersten “Vogel-Versuche” à la Archaeopteryx! Kuckucksverwandte? Keiner der bis hier erwähnten Vögel ist mit den Kuckucksvögeln verwandt …, davon abgesehen sind tatsächlich einige Kuckucksvögel sehr wohl Zugvögel, nämlich all die Arten, die in den gemäßigten Zonen der Erde leben, und wieso sollten diese Vögel Ozeane überqueren, wenn es genügend Landbrücken gab oder gibt, die die Kontinente miteinander verbinden?
***
“In the LRT giant lightless [sic] South American terror birds are more closely related to African secretary birds (Sagittarius). Traditionally Cariama has been cherry-picked for that role, but Cariama is closer to flamingos and lyrebirds …. According to results recovered by the LRT the phylogenetic split of secretary birds from terror birds must have also occurred before the appearance of the Atlantic Ocean. So in the Early Cretaceous of South America we should expect to find secretary bird bones someday. The value of this hypothesis, like any hypothesis, can be measured by its predictive value. Let’s see what happens.“
Übersetzung:
“Im LRT sind riesige flugunfähige südamerikanische Terrorvögel näher mit afrikanischen Sekretären (Sagittarius) verwandt. Traditionell wurde Cariama für diese Rolle ausgewählt, aber Cariama steht Flamingos und Leierschwänzen näher …. Nach den vom LRT gewonnenen Ergebnissen muss die phylogenetische Trennung von Sekretärvögeln und Terrorvögeln auch vor dem Erscheinen des Atlantischen Ozeans stattgefunden haben. In der frühen Kreidezeit Südamerikas sollten wir also damit rechnen, eines Tages Sekretärknochen zu finden. Der Wert dieser Hypothese kann wie jede Hypothese an ihrem Vorhersagewert gemessen werden. Mal sehen was passiert.“
Nein, der Sekretär ist nicht mit den Seriemas verwandt, beide haben nur ähnliche Lebensweisen und weisen daher einen ähnlichen Körperbau auf, dies nennt man konvergente Evolution, ein Konzept, dass D. P. nicht begreift, was er wirklich immer und immer wieder unter Beweis stellt! Auch diese beiden Vogelgruppen gab es noch nicht in der Kreidezeit, erst recht nicht in der unteren Kreidezeit, sie können sich also ebenfalls nicht während dieser Zeit aufgespalten haben! Niemand wird in Unterkreide-Lagerstätten Südamerikas Knochen von Sekretären finden, es wäre schon absolut verwunderlich überhaupt Knochen von Neornithes zu finden, da deren erste Vertreter nämlich überhaupt erst in der Oberkreide auftauchen. Der Wert dieser Hypothese, wenn es denn überhaupt eine ist, ist gleich Null.
Der Blaubrustpipra ist ein Vertreter der suboscinen Sperlingsvögel, also der Unterordnung Tyranni, er ist somit ein Vertreter der vermutlich zweitjüngsten der drei heute noch existierenden Unterordnungen der Sperlingsvögel, er ist nicht mit den Kuckucksvögeln verwandt, und dies gilt erst recht für alle anderen der oben genannten Arten, Gattungen und Familien.
*********************
bearbeitet: 05.05.2022
Heckenbraunelle

*********************
bearbeitet: 10.04.2022
